
Fluid Overload
Diagnosis and Management
- 252 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
Fluid Overload
Diagnosis and Management
About this book
Fluid overload is often observed in patients with heart failure and secondary oliguric states. A thorough assessment of the fluid status of the patient may help guide the therapy and prevent complications induced by inappropriate therapeutic strategies.The present publication is divided into four parts: Definition and Classification, Pathophysiology, Diagnosis and Therapy. In the first section, the authors present new definitions for heart failure, acute kidney injury and cardiorenal syndromes to facilitate the process of understanding the complex link between the heart and the kidney. Subsequently, different conditions leading to fluid overload are described, followed by an account of emerging diagnostic tools, therapies and technologies devoted to the treatment of patients with severe fluid-related disorders. Clearly structured and written, the present book is a practical tool for physicians and professionals involved in the management and care of patients with combined heart and kidney disorders. Moreover, it also serves as a reference textbook for medical students, residents and fellows dealing in everyday practice with fluid overloaded and oliguric patients.
Tools to learn more effectively

Saving Books

Keyword Search

Annotating Text

Listen to it instead
Information
Contrib Nephrol. Basel, Karger, 2010, vol 164, pp 88–117
Use of Biomarkers in Evaluation of Patients with Heart Failure
Abstract
Health-care-related features | Test-related features |
Give objectivity to evaluation and diagnosis of patients, especially those in whom signs and symptoms are not very sensitive or specific | High sensitivity and specificity |
Help with correct triage by assessment of prognosis and risk stratification | Reproducibility and accuracy |
Help to objectively guide the therapy or management | Low coefficient of variation |
Enable an optimum screening process | Easy to perform and analyze, possibly a point of care test |
Guide the delivery of cost-effective medical care | Applicable across sexes, ethnicity and age spectra |
Make physiological sense | |
Adapted from Maisel etal. [2]. |
Biomarkers in Heart Failure
Introduction
Markers of Myocardial Stretch
Natriuretic Peptides
Biology
NP | Actions |
ANP | arterial vasodilation, venodilation, natriuresis, diuresis, antagonizes reninaldosterone-angiotensin system, increases renal GFR |
BNP | arterial vasodilation, venodilation, natriuresis, diuresis, antagonizes reninaldosterone-angiotensin system, antiproliferative |
CNP | arterial vasodilation, venodilation, natriuresis, diuresis, antagonizes reninaldosterone-angiotensin system, antiproliferative |
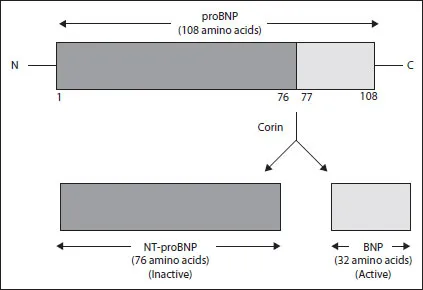
Natriuretic Peptides in Acute Heart Failure
Table of contents
- Cover Page
- Front Matter
- Heart Failure: Pathophysiology and Clinical Picture
- Heart Failure Classifications – Guidelines
- Acute Kidney Injury: Classification and Staging
- Cardiorenal Syndromes: Definition and Classification
- Oliguria and Fluid Overload
- Pathophysiology of Fluid Retention in Heart Failure
- Fluid Overload as a Biomarker of Heart Failure and Acute Kidney Injury
- Fluid Balance Issues in the Critically Ill Patient
- Prerenal Azotemia in Congestive Heart Failure
- Use of Biomarkers in Evaluation of Patients with Heart Failure
- Oliguria, Creatinine and Other Biomarkers of Acute Kidney Injury
- Current Techniques of Fluid Status Assessment
- Bioelectric Impedance Measurement for Fluid Status Assessment
- Diuretic Therapy in Fluid-Overloaded and Heart Failure Patients
- Pharmacological Therapy of Cardiorenal Syndromes and Heart Failure
- Extracorporeal Fluid Removal in Heart Failure Patients
- Technical Aspects of Extracorporeal Ultrafiltration: Mechanisms, Monitoring and Dedicated Technology
- Use of Brain Natriuretic Peptide and Bioimpedance to Guide Therapy in Heart Failure Patients
- Fluid Management in Pediatric Intensive Care
- Fluid Assessment and Management in the Emergency Department
- Author Index
- Subject Index
Frequently asked questions
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app