Asthma, allergy and chronic obstructive lung disease are common throughout the world and are increasing in incidence, particularly in the developing world. This volume provides a state-of-the-art account of the identification of new targets and the development of new therapies for these conditions. Some 40 chapters by clinical academics and senior members of the pharmaceutical industry detail the latest breakthroughs in research and development. In asthma, a promising approach is the use of therapy directed against specific Th2 responses through biological antagonists of IL-5, IL-4 and IL-13. There have also been major advances in our understanding of innate immune responses to pathogen-associated molecular patterns, and in the area of Toll-like receptors.
Up to date and comprehensive, this book will be of particular relevance to those working in the pharmaceutical industry (in preclinical research and clinical development), to academic researchers in the field of respiratory medicine, and to respiratory health care specialists.

- 324 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
New Drugs and Targets for Asthma and COPD
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
MedicineSubtopic
ImmunologyCytokines
Hansel TT, Barnes PJ (eds): New Drugs and Targets for Asthma and COPD.
Prog Respir Res. Basel, Karger 2010, vol 39, pp 103– 107
Prog Respir Res. Basel, Karger 2010, vol 39, pp 103– 107
IL-5-directed approaches in the treatment of eosinophil-driven disease
Marshall Richard,
GlaxoSmithKline Stevenage UK
Isidore Faiferman
GlaxoSmithKline King of Prussia Pennsylvania USA
Eosinophils are involved in the initiation and propagation of diverse inflammatory responses that may result in tissue damage and dysfunction. A number of diseases such as eosinophilic bronchitis, hypereosinophilic syndrome, eosinophilic esophagitis and eosinophilic gastroenteritis are currently considered to be predominantly eosinophil-driven. In atopic disease, their role has been less clear. Eosinophils are critically dependent on the cytokine interleukin-5 (IL-5), for their maturation in bone marrow, and also influence eosinophil migration and survival. Recent clinical data with monoclonal antibodies (mAbs) directed against IL-5 support this assumption. Mepolizumab (SB-240563, GlaxoSmithKline) is a humanized mouse anti-human IL-5 mAb and has been shown to be safe and effective in the treatment of patients with hypereosinophilic syndrome. Most recently, clinical trials in severe asthma and nasal polyposis have also reported positive data increasing our understanding of the role eosinophils play in these disorders.
The targets
Eosinophils are involved in the initiation and propagation of diverse inflammatory responses, including antigen presentation and the release of proinflammatory cytokines, chemokines, lipid mediators and cytotoxic granules (1-4) (Fig. 1). Tissue damage and dysfunction resulting from secretion of these pleiotropic mediators is a common pathogenic component in several diseases, including atopic asthma (5), eosinophilic bronchitis (6), nasal polyposis (7), atopic dermatitis (8, 9), eosinophil-associated gastrointestinal disorders (10) (eosinophilic esophagitis [EE], eosinophilic gastroenteritis) and hypereosinophilic syndromes (HES) (11).
Interleukin (IL)-3, IL-5 and granulocyte-macrophage colony-stimulating factor (GM-CSF) are involved in regulating eosinophil development. Of these, IL-5 is most specific to eosinophils; IL-5 is the major hematopoietin responsible for eosinophil growth and differentiation (1-4). In humans, IL-5 activity appears to be restricted to the eosinophil-basophil lineages. The IL-5 receptor complex expressed on the cell surface comprises two components: the α -chain specific for IL-5, and the β -chain, which is the signal-transducing unit. IL-5 receptor activation also modulates basophil function, especially histamine release (12). Thus, IL-5 inhibition should attenuate many eosinophil and basophil activities without affecting the function of other cells of the immune system (13), potentially limiting any safety issues, and making it an attractive drug target.
Two humanized monoclonal antibodies (mAbs) that bind to human IL-5 have been developed and tested in clinical trials. Mepolizumab (SB-240563, GlaxoSmithKline) is a humanized mouse monoclonal anti-human IL-5 antibody of the IgG1/κ subtype that binds to human IL-5 with high affinity and specificity, thus preventing IL-5 binding to the IL-5 receptor complex α -chain on the eosinophil cell surface (14). Reslizumab (Ception Therapeutics) is a humanized rat monoclonal anti-human IL-5 antibody of the IgG4/K subtype that neutralizes IL-5 by binding to amino acids 89-92 (15). Clinical development of mepolizumab is ongoing in hypereosinophilia, severe asthma and nasal polyposis; reslizumab has recently entered phase II/III for eosinophilic esophagitis in children. Other approaches in early development include a series of monoclonal antibodies directed against IL-5Rα (MEDI-563, MedImmune), an IL-5-based vaccine (Cytos Biotechnology) and an antisense oligo-nucleotide to the common β -chain of the IL-5 receptor (Topigen).
Pharmacology
Mepolizumab displays cross-reactivity with cynomolgus monkey IL-5; thus, these animals were used as the in vivo model for assessment of the pharmacology and long-term safety of mepolizumab. Studies in rats and mice were conducted using an anti-rat IL-5-specific mAb; this antibody cross-reacted with mouse IL-5.
Preclinical studies in rodent and primate models showed that mepolizumab produced a profound and prolonged (> 3 months) suppression of eosinophil production and prevented antigen-induced lung eosinophilia. Eosinophil counts were taken as a proxy measure of mepolizumab pharmaco-dynamics. Peripheral eosinophil count decreased in a time-dependent manner following a single subcutaneous injection of mepolizumab. The maximal response (80-95% decrease in eosinophil count compared to baseline) occurred at 3-4 weeks post-dosing. Following 6-monthly repeated administrations of mepolizumab (10 mg/kg) the observed eosinophil counts were persistently < 20% of the baseline levels over the entire 6-month dosing period. The estimated IC50 value of mepolizumab for reducing the levels of circulating eosinophils was 1.43 µ g/ml. Multiple dose administrations did not produce the expected degree of reduction as predicted by the pharmacodynamic single-dose model (16).
Pharmacokinetic profile
Following i.v. administration to cynomolgus monkeys, mepolizumab exhibited a relatively low clearance and volume of distribution. Mepolizumab concentration declined in a biexponential manner, with a mean half-life (t1/2) of 12.9 ± 9.4 h. The mean overall terminal t1/2 following intravenous dosing was 13 ± 2 days. After subcutaneous dosing, mepolizumab was completely absorbed into the systemic circulation and appeared to decline in a mono-exponential manner with an apparent t1/2 of 14.5 ± 3.8 days. Maximal concentrations were observed 2 to 4 days postinjection. Studies in monkeys did not reveal any anti-mepolizumab antibody following repeated i.v. or s.c. administration. The mean half-life of mepolizumab given i.v. to patients with asthma was 20 ± 2.5 days (16).
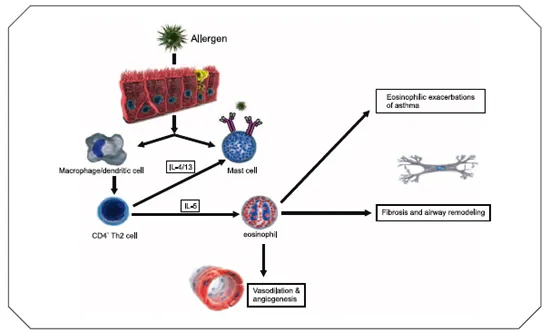
Figure 1. Biology and actions of interleukin-5 (IL-5).
Preclinical animal models
In a cynomolgus monkey model of IL-5-dependent eosinophilia, s.c. administration of 6 doses (1/day on alternate days) of recombinant human IL-2 produced a profound eosinophilia. This eosinophilia was blocked by ≥ 85% (p < 0.05 vs. untreated controls) in monkeys pretreated with mepolizumab (2 i.v. doses of 0.5, 5 or 50 mg/kg at 1 and 29 days before IL-2 administration) (14).
A single 10 mg/kg i.v. dose of mepolizumab in Ascaris suum-sensitive monkeys (n = 8) significantly decreased the number of eosinophils in blood following A. suum challenge at day 1 and week 6 (p < 0.05 vs. control-treated animals) and the number of eosinophils in the bronchoalveolar lavage fluid (BALF) at week 3 (p < 0.05 vs. control-treated animals) (14). The levels of regulated on activation, normal T expressed and secreted (RANTES) and IL-6, both eosinophil secretion products, in BALF at 3 weeks postdose were also decreased. However, there was no effect on the acute broncho-constrictor response to A. suum antigen challenge (14).
These studies in cynomolgus monkeys indicate that mepolizumab is safe and well tolerated in an in vivo animal model, and, by producing long-term reductions in circulating and tissue eosinophils, has therapeutic potential in diseases where overproduction of eosinophils plays an important pathological role.
Experimental murine models of EE suggest a major role for IL-5 in the pathogenesis of this disease. EE can be induced by overexpression of IL-5 in transgenic mice (10) and blocked by neutralizing IL-5 in an allergen-induced murine model of EE (17). Furthermore, mice deficient in IL-5 are protected from induction of experimental EE (17, 18).
Human clinical studies
Asthma
Initial clinical studies confirmed the safety profile, and the efficacy in reducing circulating eosinophils seen in preclinical species, but called into question the relevance of eosinophils in asthma. Single intravenous infusions of mepolizumab (2.5 mg/kg and 10.0 mg/kg) in patients with mild allergic asthma reduced blood eosinophils by about 85% versus baseline or placebo; this reduction lasted for 16 weeks for the higher dose of mAb used (19). In a subsequent study, patients with mild atopic asthma received mepolizumab 750 mg (n = 11) or placebo (n = 13) monthly for 3 months. Mepolizumab reduced blood eosinophils by 100% and there was a median reduction in BALF eosinophils of 80% . On the other hand, there was a median reduction of only 55% in airway mucosal MBP+ eosinophils. There were no significant changes in FEV1 or PEFR (peak expiratory flow rate), or in airway basophils, neutrophils, macrophages, mast cells or CD3+ cells (20).
In another study, patients with symptomatic asthma of moderate severity, taking ≤ 1,000 µ g/day beclomethasone or equivalent, received mepolizumab 250 mg (n = 120) or 750 mg (n = 116) or placebo (n = 126) once a month, for 3 months (week 12), and were followed for an additional 2 months (week 20). There was a statistically significant reduction in blood and sputum eosinophil counts for the duration of the study, including follow-up; there were no...
Table of contents
- Cover Page
- Front Matter
- Introduction
- Bronchodilators
- IgE- and epithelial-directed approaches
- T-cell co-stimulator blockade
- Pattern recognition receptors
- Cytokines
- Chemokines
- Adhesion molecules
- Mediator receptor antagonists
- Enzyme inhibitors
- Antioxidants
- Protease inhibitors
- Inhibitors of fibrosis and vessels
- Inhibitors of cell signaling and transcription
- Nucleic acid therapy
- Author Index
- Subject Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access New Drugs and Targets for Asthma and COPD by T. T. Hansel,P. J. Barnes, J. J. F. Herth in PDF and/or ePUB format, as well as other popular books in Medicine & Immunology. We have over 1.5 million books available in our catalogue for you to explore.