![]()
CHAPTER 1
Applications of Smart
Multifunctional Tissue
Engineering Scaffolds
M. KALIVAa,b, M. CHATZINIKOLAIDOUa,b AND M. VAMVAKAKI*a,b
a Department of Materials Science and Technology, University of Crete, P.O. Box 2208, 710 03, Heraklion, Crete, Greece;
b Institute of Electronic Structure and Laser, Foundation for Research and Technology-Hellas, P.O. Box 1527, 711 10, Heraklion, Crete, Greece
1.1 Introduction
Tissue engineering is an attractive approach to restore and replace diseased or defective tissue offering an alternative to other clinical methods such as organ replacement. Conventional tissue engineering approaches involve the use of a scaffold mainly as a structural element with defined physicochemical, mechanical and biological properties and appropriate architecture and porosity to support cell metabolism. However, recent approaches in tissue regeneration combine three key elements: a scaffold as a micro-environment to promote cell adhesion for tissue development, an appropriate cell type, and biomolecules and drugs to guide cell response and function.1–3 There has been enormous interest lately in the growth of different types of tissues using multifunctional scaffolds that can actively participate in the process to provide the biological signals that guide and direct cell function (proliferation, growth and differentiation). Such scaffolds are derived from novel functional and smart materials that allow tuning of the properties and behavior of the scaffolds and can perform multiple crucial tasks simultaneously i.e. deliver bioactive and pharmaceutical molecules, direct cell growth and differentiation, and control stem cell behavior.4
Organic, inorganic and hybrid (organic–inorganic) materials have all been explored in the development of multifunctional scaffolds. Basic material requirements for use in tissue engineering include biocompatibility, histocompatibility, non-toxicity and the ability to engineer an appropriate scaffold with the required functionalities.
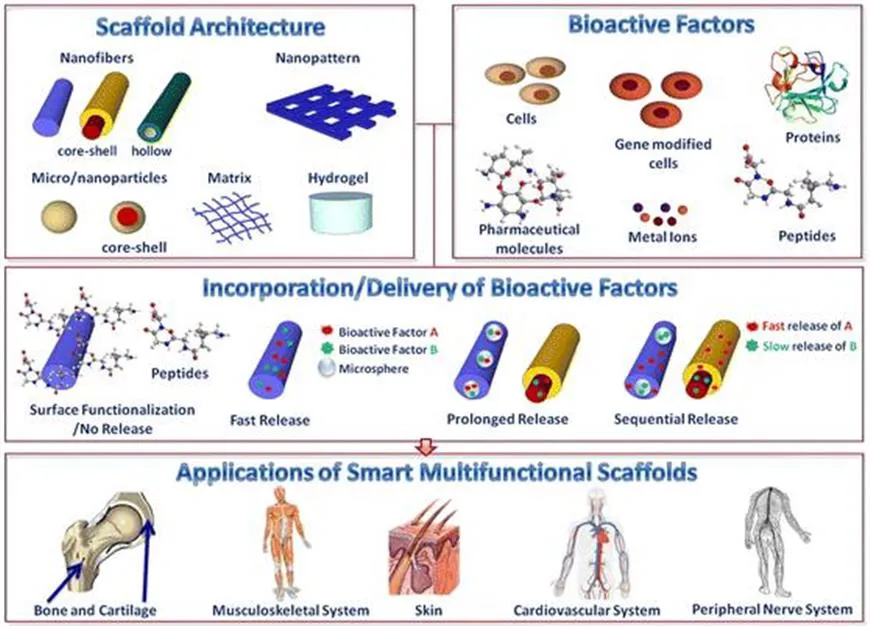
Multifunctional scaffolds based on smart materials have been applied in different tissue engineering fields. The most frequently studied areas in the literature include the use of multifunctional scaffolds in bone, cartilage and muscle formation, in cardiovascular and endothelium tissue engineering, in the growth of skin and in neural regeneration. Other applications include their use in dental, corneal and retina tissue engineering as well as in wound healing. This chapter will focus on the most extensively studied tissues of which the understanding and knowledge have matured the most. Although multifunctional materials and stimuli-sensitive nanoparticulate drug delivery systems have also shown great therapeutic potential for various cardiovascular and infectious diseases and cancer,5 this application will not be discussed here. In the following, the sections are divided based on the respective tissue of interest, for which the material characteristics and the multifunctionality of materials and scaffolds are discussed (Figure 1.1). The potential clinical applications of the multifunctional scaffolds are also considered.
Figure 1.1 Multifunctional materials and strategies for applications in tissue engineering.
1.2 Applications of Multifunctional Scaffolds in Tissue Engineering
1.2.1 Bone and Cartilage
Bone is a remarkably organized, hierarchical connective and vascularized tissue that provides mechanical support and serves various biological functions. Degenerative diseases, cancer or injury can cause bone defects. Despite the impressive ability of bone to heal spontaneously after trauma or fractures, a significant need still exists to develop strategies that promote the healing of non-spontaneously healed defects as a result of sufficiently large fractures or diseases with poor healing ability (i.e. osteoporosis, cancer). Bone tissue regeneration is a physiological and complex procedure that involves a well-orchestrated participation of various bioactive molecules. Bone extracellular matrix (ECM) comprises different proteins such as collagen fibronectin (FN), osteocalcin (OC), osteopontin (OPN), and bone sialoprotein (BSP). Different bone morphogenetic proteins (BMPs) and growth factors, like transforming growth factor-beta (TGF-β), insulin-like growth factor (IGF), platelet-derived growth factor (PDGF), fibroblast growth factor (FGF), and vascular endothelial growth factor (VEGF), are actively involved in the process of bone regeneration, in a spatiotemporal and concentration-controlled manner.6–8 Multifunctional scaffolds, based on smart materials, are capable of promoting new bone formation, and have received particular attention in the field of bone tissue engineering lately. These scaffolds must indulge a series of different requirements such as bioactivity, biocompatibility, controllable biodegradability, appropriate mechanical strength, architecture and porosity, sustained delivery of chemical and biological cues (growth factors, genes, peptides, small bioactive molecules and ions) to eliminate infection from pathogens and reduce immune response, while promoting cell attachment and growth and stimulating osteo-differentiation and angiogenesis.9–12
1.2.1.1 Natural Polymers
Natural polymers have been extensively employed as multifunctional materials in bone and cartilage tissue engineering. This is driven by their superior biological response and their behavior that closely mimics tissue replacement, as well as the inherent non-toxicity and biodegradability of these materials, which renders them particularly attractive for use in biomedical applications. Silk possesses good mechanical properties that can be combined with the adhesive properties of the tripeptide Arg-Gly-Asp (RGD) for the development of robust multifunctional scaffolds exhibiting good cell adhesive properties, high wettability and enhanced biodegradability supporting the attachment, proliferation, and spreading of MC3T3-E1 cells.13
Enormous effort has been focused on the potential of cells and stem cells to differentiate because it allows the growth of tissues in vitro before their implantation in vivo using a variety of available cells. The design of the scaffold microenvironment, along with the presentation of appropriate cues to induce the differentiation of stem cells, is a highly promising strategy in tissue engineering. The surface functionalization of biomaterial scaffolds with biomimetic proteins is commonly employed in this direction. Collagen is the most frequently used matrix as it is found in the extracellular matrix and provides mechanical strength and supports bone formation. Collagen-based scaffolds have been shown to increase the adhesion, growth, and differentiation of osteoblastic cells and promote tissue formation in vivo. On the other hand, adhesive molecules such as FN can regulate cellular recognition of the scaffold through integrin signaling. OC, a non-collagenous protein with a high affinity for mineral crystals, promotes the biomineralization process and has been reported as the key factor during the late phase of osteoblasts and stem cell differentiation. In a novel strategy, OC-FN possessing a collagen binding domain has been integrated in a collagen fibrillar network to provide multifunctional and highly stable scaffolds.14 The FN active sites enhance the attachment of mesenchymal stem cells (MSC) onto the hybrid matrix, whereas a rapid cell confluence and differentiation to a mature and osteogenic phenotype is driven by OC, leading to significantly improved in vivo bone formation in calvarial defects.
BMPs are an important protein family used extensively for the differentiation of pre-osteoblasts and MSCs into osteogenic and chondrogenic cells in bone and cartilage tissue engineering. Among them, BMP-2 exhibits high osteoinductive capacity.15 However, the delivery mode of BMP-2 from the carrier affects the efficacy of bone regeneration. A sustained in vivo delivery of BMP-2 has been shown to favour bone formation compared to the burst release of the protein.16,17 Site specific binding and regulated delivery of BMP-2 can prolong its delivery and maintain a higher local concentration at the bone injury site. Vehicles based on different biomaterials have been used to tackle this challenge, among which, demineralized bone matrix collagen, derived from cancellous bone tissues, is particularly attractive because it resembles the human bone structure and composition. The specific conjugation of a monoclonal antibody containing six histidine tags on the collagen scaffold followed by BMP-2 binding using orthogonal chemistries increases the loading capacity of the scaffold for BMP-2 and its ability to control the release in vitro.18 The multifunctional scaffolds show increased osteogenic differentiation due to the presence of BMP-2 and more ectopic bone formation. Another strategy employs multifunctional porous or nanoparticulate materials that can deliver single or multiple growth factors in a controllable manner. Alginate is a particularly attractive matrix due to its inertness and lack of interference in the signaling molecules–cells interactions. Macro-porous alginate scaffolds functionalized with both the TGF-β1 chondrogenic-inducing factor and the RGD peptide strongly affect the MSC morphology, viability and proliferation as well as cell differentiation and the appearance of committed chondrocytes, leading to more effective chondrogenesis compared to the scaffolds functionalized solely with TGF-β1.19 This is attributed to the effective cell–matrix interactions promoted by the immobilized RGD peptide, which result in a better cell accessibility to the TG-Fβ1 inducer. The regulatory role of TGF-β1 in the osteogenic activity of BMP-2 has been further confirmed in collagen sponge scaffolds. Regulation of the osteoblast and osteoclast gen...