![]()
1
Physiology of the Blood
and Bone Marrow
What Do You Have to Know?
Which cells are normally present in the blood and what is their function
The normal intravascular life span of erythrocytes, neutrophils and platelets
Where and how blood cells are produced
The production and function of erythropoietin
The source and absorption of iron, vitamin B
12 and folic acid, and their role in haemopoiesis
Blood Cells and Their Functions
The circulating blood contains red cells, white cells and platelets suspended in plasma (Fig. 1.1). The cells in the circulating blood originate in the bone marrow.
Red cells, also known as erythrocytes, differ from most body cells in that they no longer have a nucleus. This is extruded when they leave the bone marrow. Normal mature red cells are disc-shaped and, because they lack a nucleus, are flexible; they can deform and squeeze though capillaries. The major function of the red cell is the transport of oxygen from the lungs to peripheral tissues but it also transports CO2 from the tissues to the lungs and has a role in nitric oxide (NO) transport and metabolism, favouring generation of NO and vasodilation in conditions of hypoxia.
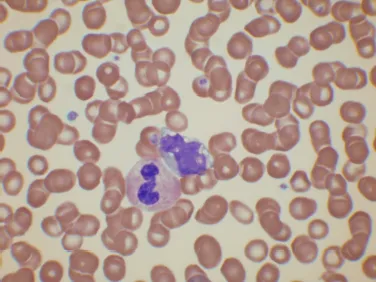
Fig. 1.1. A stained blood film showing erythrocytes, leucocytes (a neutrophil and a monocyte) and platelets.
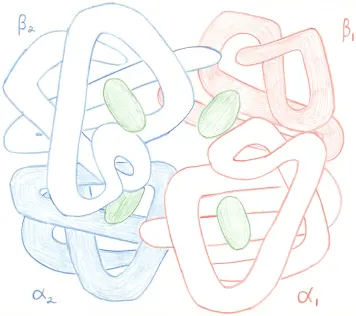
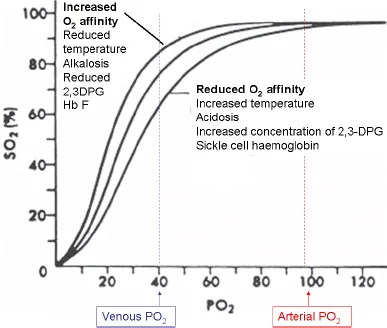
The principal constituent of the red cell, responsible for oxygen transport and contributing to CO2 transport and interactions with NO, is haemoglobin. It is an iron-containing protein composed of four polypeptide chains known as globin chains, each of which has a deep pocket into which an iron-containing haem group is inserted. The tetrameric haemoglobin molecule is composed of two heterodimers: in normal adult haemoglobin (haemoglobin A) the dimer is composed of an α and a β chain (Fig. 1.2). Oxygen for transport can enter the haem pocket and bind reversibly to haem. The globin chains can alter their relationship to each other in response to uptake or release of oxygen by haem. The loss of the oxygen molecule from one haem makes it more likely that other oxygen molecules will be lost from other haems. This is known as co-operativity and is responsible for the sigmoid shape of the oxygen dissociation curve (Fig. 1.3). Co-operativity ensures that haemoglobin takes up oxygen readily as it passes through the lungs, becoming almost fully saturated but, when it reaches tissues, oxygen is equally readily released. Delivery of oxygen in the tissues is facilitated by a higher temperature and a lower pH so that more oxygen is available to metabolically-active tissues. It is also facilitated by interaction with 2,3 diphosphoglycerate (2,3 DPG), a glycolytic intermediate that increases in concentration in response to anaemia. Higher temperature, lower pH and a higher concentration of 2,3 DPG thus decrease the affinity of haemoglobin for oxygen.
Fig. 1.2. A diagram of a haemoglobin molecule, showing two α and two β globin chains, each with a haem in the haem pocket. The α1β1 dimer is shown in pink and the β2β2 dimer in blue. The haem group is represented in green.
Fig. 1.3. The oxygen dissociation curve illustrating the sigmoid curve that results from co-operativity.
Erythrocytes are enclosed by a surface membrane, which is a lipid bilayer, supported by a cytoskeleton that maintains the biconcave shape of the cell. They contain enzymes of the Emden-Meyerhoff (or glycolytic) pathway, which meets the energy needs of the cell and enzymes of the pentose (hexosemonophosphate) shunt, which protects the cell from oxidant damage. The erythrocyte life span is about 120 days, effete cells being removed by macrophages, particularly in the spleen. The synthesis of haemoglobin is discussed further in Chapter 5 and the red cell membrane and enzymes in Chapter 6.
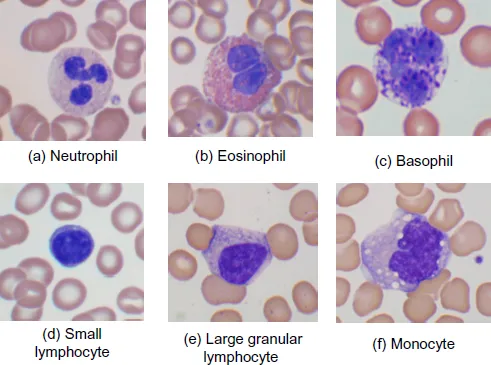
White cells, also known as leucocytes, defend the body against infection and participate in immune responses. Those that are normally present in the blood are of five types. Three of these are referred to as granulocytes, because their cytoplasm contains granules; depending on the colour of the granules in a stained blood film they can be divided into neutrophils (small lilac granules), eosinophils (larger orange granules) and basophils (large purple granules). They all have lobulated or polymorphous nuclei and thus are sometimes referred to as polymorphonuclear leucocytes or ‘polymorphs.’ Granulocytes function mainly in the tissues, rather than in the bloodstream. They reach tissues by migrating through the endothelium of capillaries.
Neutrophils (Fig. 1.4a) spend only about 7 hours in the circulation. They are phagocytic cells that respond to chemotactic stimuli by migrating to sites of infection, inflammation or cell death. This process involves rolling along the endothelium, adhering to specific endothelial receptors, moving through capillary walls (diapedesis) and migrating through tissues in response to chemotaxins. The neutrophils engulf bacteria and other unwanted material within tissues by a process known as phagocytosis. This involves flowing of pseudopodia around the particle with subsequent fusion so that the unwanted bacterium or other particle is enclosed in a phagocytic vacuole within the cytoplasm. The granules of the neutrophil (which contain proteolytic enzymes and myeloperoxidase) are then discharged into the phagocytic vacuole, where H2O2 and other reactive oxygen species are generated. The result is killing of microbes and proteolysis of phagosome contents. Neutrophils spend about 30 hours in tissues.
Fig. 1.4. Normal leucocytes in the peripheral blood: (a) a neutrophil; (b) an eosinophil; (c) a basophil; (d) a small lymphocyte; (e) a large granular lymphocyte; (f) a monocyte.
The major function of eosinophils (Fig. 1.4b) is defence against parasitic infection. They are less efficient than neutrophils in defence against bacteria. In addition to these beneficial functions, eosinophils have unwanted actions when they are involved in allergic reactions. Basophils (Fig. 1.4c) also participate in defence against parasites and allergic responses.
Lymphocytes (Figs. 1.4d, e) are smaller than granulocytes and have a round nucleus. A minority of them have a small number of cytoplasmic granules. Circulating lymphocytes look very similar to each other but include cells of three lineages, B cells, T cells and natural killer (NK) cells. B cells are of bone marrow origin. They migrate from the bloodstream to lymph nodes or other lymphoid tissues where they are exposed to antigens presented to them by dendritic cells (antigen-presenting cells). Their exit from the bloodstream is through high endothelial venules of lymph nodes and post-capillary venules of other tissues. Antigen exposure leads to somatic mutation so that cells most capable of recognising the antigen survive and mature into both memory B cells and antibody-secreting plasma cells, responsible for humoral immunity.
T cells are also of bone marrow origin but have undergone maturation in the thymus. After thymic maturation they migrate to lymph nodes and other lymphoid tissues. There are diverse subsets of T cells. Some function in cell-mediated immune responses, binding to and damaging antibody-coated cells or microorganisms (cytotoxic T cells). They also modulate the function of B cells, acting as helper or suppressor cells, activate macrophages and attract and activate neutrophils. Some resemble NK cells in that they have cytotoxic effect without the need for recognition of an antigen. There is also a subset of T cells that has a regulatory function, including maintaining immune tolerance.
NK cells are part of the body’s innate immune response, being able to attack cancer cells and foreign cells even though they are not antibody-coated. Lymphocytes that contain granules are either NK cells or cytotoxic T cells. Otherwise it is not possible to distinguish T cells from B cells by their appearance in the blood film. Lymphocytes recirculate between the lymphatic system and the bloodstream. They spend a very variable period of time in the circulation. Their survival is very variable but in some cases this may amount to many years.
Monocytes (Fig. 1.4f) are the largest cells normally present in the blood. They have a lobulated nucleus and plentiful cytoplasm. They spend several days in the circulation but their main function is in the tissues. There they mature into macrophages or histiocytes (collectively known as the reticuloendothelial system), capable of phagocytosing and killing microorganisms and breaking down and removing cellular debris. They present antigens to lymphocytes. Whereas neutrophils are most important in defence against acute bacterial infection, cells of the monocyte/macrophage lineage are most important in defence against chronic bacterial infections, such as tuberculosis, and in chronic fungal infections. They also secrete numerous cytokines that enhance the inflammatory response to infection and also growth factors that promote the production of neutrophils and monocytes. In addition, macrophages remove parasites (such as malaria parasites) and other particles from red cells. They destroy red cells at the end of their life span and store the iron released from haemoglobin so that it can be recycled.
Platelets are small particles formed by fragmentation of the cytoplasm of bone marrow megakaryocytes. They function in the primary haemostatic response, forming a platelet plug at the site of small vessel injury. When activated, they also expose altered phospholipid on their surfaces that interacts with coagulation factors to promote blood coagulation at the site of tissue injury. They survive for about 10 days in the circulation.
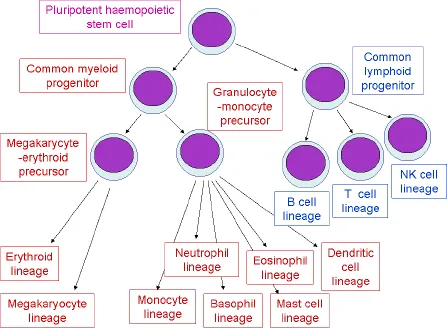
Fig. 1.5. The haemopoietic stem cell hierarchy.
Bone Marrow Cells and Normal Haemopoiesis
All the blood cells are ultimately derived, in the bone marrow, from a pluripotent haemopoietic stem cell that is capable of giving rise to both lymphoid and myeloid progeny via a common lymphoid progenitor cell and a common myeloid progenitor cell respectively (Fig. 1.5). The common lymphoid progenitor gives rise to B cells, T cells and NK cells. The common myeloid progenitor is a multipotent haemopoietic progenitor cell (sometimes also known as a multipotent myeloid stem cell) capable of giving rise to cells of all myeloid lineages. It gives rise initially to an erythroid-megakaryocyte progenitor cell and to a granulocyte-monocyte progenitor cell. The granulocyte-monocyte progenitor gives rise not only to monocytes and the three types of granulocyte, but also to the antigen-presenting dendritic cells and to mast cells, which are tissue cells concerned with immune responses. The monocyte/macrophage lineage gives rise to many specialised tissue macrophages (e.g. Kupffer cells in the live...