![]()
CHAPTER 1
THE PARTICULATE NATURE
OF MATTER
1.1What Is Chemistry?
In order to know what chemistry is all about, firstly, it is important to know that the Universe is made up of matter. What is matter? Things that have mass and take up space are known as matter.
Chemistry is an important branch of science that concerns with the study of the properties of matter and how different matters react (known as reactant) to give one or more substances (known as product) which have different physical and chemical properties from the reactants.
QWhat is a physical property?
A:The physical property of a matter is an observable and measureable quantity used to describe the matter. Physical property can be changed without changing the basic composition of the matter. Examples include temperature, pressure, density, mass, volume, color, boiling point, melting point, energy content, and many more. The melting of ice is a change in the physical property of water. Such physical changes are reversible by changing the conditions such as temperature and pressure back to original.
QWhat is a chemical property?
A:As different types of matter have different chemical compositions (we would later learn the different kinds of fundamental particles that different types of matter are made up of in Chapter 3 under Chemical Bonding), each matter has its own unique potential to react with other matter. This unique potential to undergo chemical change is known as the chemical property of a matter. For example, a piece of paper has the potential to be ignited by a flame. This is a chemical change and during this change new substances such as carbon dioxide and water are formed. Such chemical changes are usually irreversible by any simple change of conditions.
1.2States of Matter
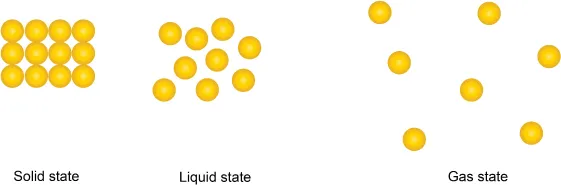
As matter is made up of very small particles (we would later learn that the particles can be atom, ion, or molecule) being attracted to one another. The different strengths of the attraction between these small particles result in different physical states of matter.
If the strength of attraction is very strong, we have solid matter, which gives rise to its fixed shape and fixed volume. A weaker strength of attraction gives us liquid, which has a fixed volume but no fixed shape. The strength of attraction is the weakest in a gas, causing it to have neither a fixed shape nor a fixed volume.
QSo in the solid state, the particles are so close to one another that the boundaries of the particles are “touching” each other?
A:No, this is not true! In fact, if you can go down to the microscopic or atomic level (we would see what an atom means later), there is actually still a huge gap between the particles. And in between these gaps, it is just vacuum! There is nothing else in between.
QWhat is the force that caused the small particles to be attracted to one another?
A:The force that caused the small particles to be attracted together is known as electrostatic force. Do not fret, it is just a type of “electrical force” between particles that possess opposite “electrical charges.” We would learn this in more detail in Chapter 3 on Chemical Bonding, as such attractive forces are the basis for chemical bonds that hold the particles together.
QSo, the fact that matter has different states is the result of chemical bonds of different strengths?
A:Yes, you are right. Because of the chemical bonds of different strengths, it results in different arrangements of particles in the respective physical states, which would decide whether the matter has a fixed shape or volume. And because of the different types of arrangement, the particles have different types of motion in the physical states and different levels of compressibility. So, it is also because when different states of matter possessing different chemical bond strengths react, different amounts of energy are involved in the chemical reaction. We would learn more about this in Chapter 5 on Energy Change and Fuels.
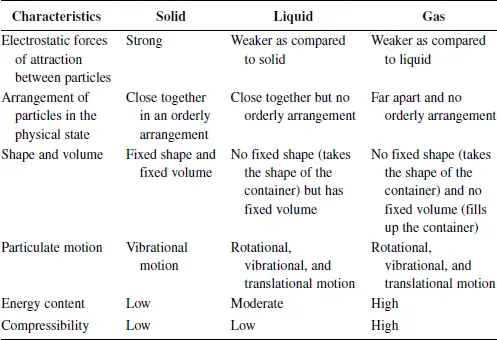
Summary of important characteristics for the states of matter
QWhat is the difference between rotational, vibrational, and translational motions?
A:Rotation is movement ABOUT an axis, vibration is movement ALONG an axis, andtranslation is movement ALONG MORE THAN ONE axes. So, translation can be movement on a two-dimensional plane or a three-dimensional space.
QWhy is the energy content of a solid lower than that of liquid, while a liquid is lower than that of a gas?
A:Take for instance, when ice melts, does it absorb energy? Yes, so what is the absorbed energy used for? As we now know that the bond strengths of matters of different physical states are the strongest between particles in the solid state, followed by liquid then gas. The energy that is absorbed when a solid melts is to “help” weaken the bonds between the particles in the solid state. As energy cannot be created nor destroyed based on the Law of Conservation of Energy, the energy that is absorbed during melting is “transferred” as the energy content of the liquid. This thus makes the liquid possess a higher energy content than the solid. Similarly, when a liquid is converted to the gaseous state, the energy content of the gas is higher than that of the liquid.
QSo, does that mean that when a liquid solidifies, the same amount of energy that is absorbed during melting is given off?
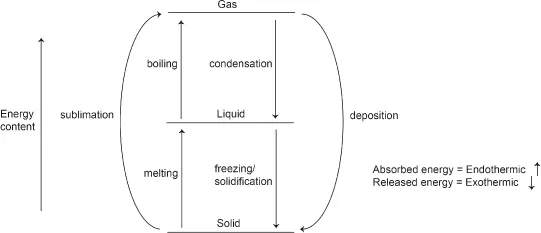
A:Absolutely spot on! The following shows the energy changes during phase changes:
The amount of energy that is involved during the forward phase change is the same as that for the reverse phase change, as shown by the same “gap” between the two different physical states.
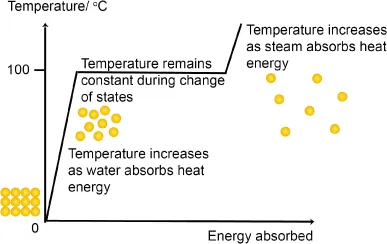
QBut since energy is absorbed during a melting process, why is the measured temperature a constant during the melting process?
A:The following is a heat curve that shows the corresponding changes in temperature versus energy absorbed (in calories) as water undergoes the phase transition between the liquid and gas states.
From 0°C to 100°C, the energy that the system has absorbed increases the temperature of the water because the energy absorbed is converted into the kinetic energy of the particles. With a higher kinetic energy, the particles move very rapidly. At the 100°C point, the kinetic energy of the particles is sufficient to help the particles overcome the attractive forces from the other particles. Hence, the particles can escape into the gas phase. Thus, the energy that is introduced into the liquid will not go into increasing the temperature anymore (as the kinetic energy of the particles ...