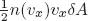-->
In this new textbook, a number of unusual applications are discussed in addition to the usual topics covered in a course on Statistical Physics. Examples are: statistical mechanics of powders, Peierls instability, graphene, Bose-Einstein condensates in a trap, Casimir effect and the quantum Hall effect. Superfluidity and super-conductivity (including the physics of high-temperature superconductors) have also been discussed extensively.
The emphasis on the treatment of these topics is pedagogic, introducing the basic tenets of statistical mechanics, with extensive and thorough discussion of the postulates, ensembles, and the relevant statistics. Many standard examples illustrate the microcanonical, canonical and grand canonical ensembles, as well as the Bose-Einstein and Fermi-Dirac statistics.
A special feature of this text is the detailed presentation of the theory of second-order phase transitions and the renormalization group, emphasizing the role of disorder. Non-equilibrium statistical physics is introduced via the Boltzmann transport equation. Additional topics covered here include metastability, glassy systems, the Langevin equation, Brownian motion, and the Fokker-Planck equation.
Graduate students will find the presentation readily accessible, since the topics have been treated with great deal of care and attention to detail.
-->
Request Inspection Copy
-->
0 Readership: Graduate students and academics focusing on statistical mechanics. -->Bose-Einstein Statistics, Fermi-Dirac Statistics, Boltzmann Transport Equation, Peierls Instability, Casimir Effect, Quantum Hall Effect, Langevin Equation, Brownian Motion, Phase Transitions0