![]()
CHAPTER 1
THE PRINCIPLE OF PHOTOELECTROCHEMICAL WATER SPLITTING
Peiyan Ma*,‡ and Dunwei Wang†,§
*School of Chemistry, Chemical Engineering and Life Science,
Wuhan University of Technology, Wuhan, China†Department of Chemistry, Boston College,
2609 Beacon St., Chestnut Hill, MA 02467, USA‡[email protected]§[email protected] Photoelectrochemical (PEC) water splitting has been attracted significant attention lately due to its utilization of solar energy and H2 production. The critical challenge in PEC research is the O2 evolution half reaction (OER) occurring on the photoanode. This chapter consists of an introduction of PEC system, the detailed process of OER, the development of several semiconductor photoanodes, OER mechanism, high-efficient elelctrocatalysts and tandem cell based on PEC system. This chapter provides a review of the principles, research route and prospect of PEC and analysis of the microstructure, interface engineering and performance in some important samples. It will be a guide for the beginners in the PEC area.
1.Introduction
1.1.General background
Excessive exploitation of fossil fuels makes it an urgent task to develop renewable and sustainable energy sources to solve challenges that come with the energy crisis. Solar energy is expected to be the main option, especially in areas with ample sunshine. Successful utilization of solar energy relies on the development of efficient harvesting materials and effective storage technologies. As far as direct solar energy conversion and storage is concerned, hydrogen (H2) has attracted significant attention owing to its high energy density (120–142 MJ/kg) and the ability to obtain it from H2O. When used as an energy carrier, H2 is highly efficient and can be used for transportation, storage, and power replacement in places where other energy sources are difficult or expensive to access. That its combustion only generates H2O is another reason why H2 is particularly appealing.
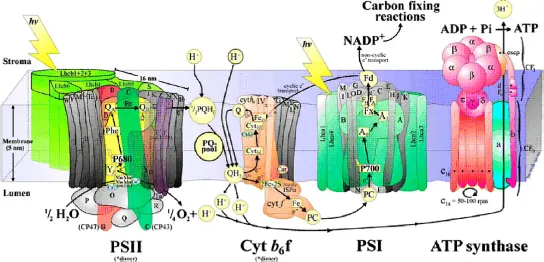
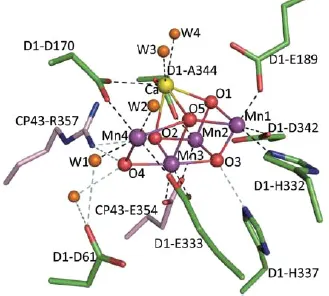
As a method developed by Mother Nature to harvest and store solar energy, photosynthesis is an important biochemical process. It produces biological oxygen molecules (O2) at the protein complex of the photosystem II (PSII) in algae, cyanobacteria and plants. Water is split to O2 and H2 or H2-related species by a two-step excitation process (Figure 1) in which the two half reactions (oxidation and reduction) are spatially separated and proceed in PSII and PSI (photosystem I), respectively [1]. The active site for this water splitting process is embedded in a protein complex 1, which consists of four manganese ions and one calcium ion (Figure 2) [2].
Figure 1. Schematic diagram of water splitting process in the membranes of the chloroplasts. Reprinted with permission from [1].
Figure 2. Structure of Mn4Ca active site in the O2 evolution catalysts (OEC). Reprinted with permission from [2].
Although it was not until 2011 that researchers successfully identify the structure of the O2 evolution center in PSII2, the natural photosynthesis route has inspired researches of a similar nature for a long time. As early as 1971, Fujishima and Honda demonstrated that TiO2 could be used to decompose water to H2 and O2 under photoelectrochemical (PEC) conditions (see below), which is widely regarded as the dawn of artificial photosynthesis research [3].
The negative value of the normal electrodynamic potential indicates that water splitting is thermodynamically uphill and additional energy is necessary to power the reaction. Relative to HER (which involves 2 electrons and 2 protons), OER is far more complex because it requires 4 electrons and 4 protons. As such, it has been identified as the rate determining step (RDS) for artificial photosynthesis. Indeed, while a number of p-type semiconductors have been shown to produce H2 with high efficiencies, n-type semiconductors for high-efficiency, stable H2O oxidation is still missing. In recognition of this critical research need, we focus on photoanode and OER reactions for this chapter.
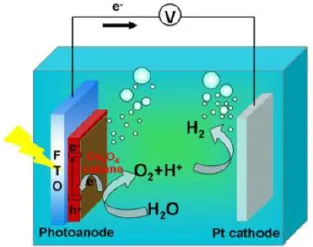
Figure 3. An PEC cell consists of a semiconductor photoanode and a Pt cathode. Cobalt-based catalyst was modified on the surface of photoanode. Reprinted with permission from [4]. Copyright @ American Chemical Society.
A typical water splitting cell device includes semiconductors with appropriate bandgap as photoelectrodes (photoanode or photocathode) to absorb sunlight, conductive substrates to transport charges and electrolyte to support the reactions (Figure 3) [4]. Electrons and holes are generated when the photoelectrodes are irradiated. They then transfer to the conduction and valence bands, respectively. The photogenerated electrons are required to flow to the surface of cathode, where they reduce H+ to H2. At the same time, photogenerated holes are expected to move to the surface of the anode and oxidize water to O2. Often, catalysts are needed to increase the reaction kinetics on both the photoanode and the photocathode surface, to match the rates of charge generation and separation within the photoelectrodes. In the following sections, we will first introduce the fundamental considerations of the semiconductor-based water splitting process, and then discuss material selection for the photoanode and end the chapter with a discussion on the detailed mechanisms.
1.2.Fundamental considerations in selecting a material for solar water splitting
After the introduction of TiO2 to water splitting by Fujishima and Honda, a large number of semiconductors have been explored for the same purpose. There are several principles for selecting a semiconductor as a photoelectrode.
1.2.1.Potential requirement
Under standard conditions, the water splitting reaction can take place only when the potential difference of the two half reactions exceeds 1.23 eV (ΔG° = 237.1 kJ·mol−1). That is, the combined bandgap of the semiconductors should be larger than 1.23 eV. Other energy losses have to be taken into account, especially the overpotential loss at the electrodes and ionic conductivity loss in the electrolyte. In a practical cell, the actual potential requirement typically exceeds 1.6 V.
1.2.2.Appropriate band structure
Aside from an appropriate bandgap, proper conduction and valance bandedge positions should be taken into consideration for water reduction and oxidation reactions, respectively. Values obtained by typical vacuum-based measurements are often not accurate due to effects such as solvent adsorption. To determine the relative positions of the bandedges, we often need experimental measurements by tools such as ultraviolet photoelectron spectroscopy (UPS), which reports on the ionization energy (effectively the energy of the valence band maximum, Ev).
1.2.3.High crystallization and surface area
In order to achieve efficient charge separation and collection, semiconductors with high crystallinity for charge transport and high surface activities for the OER/HER reactions are desired.
1.2.4.Stability
Stability is of paramount importance to the application of solar water splitting. The optimized photoelectrode material should be stable in electrolytes of a certain pH range, often in the extreme acidic or basic regime. Effective protection may be needed...