![]()
Chapter 1
Drug Delivery Systems: Possibilities and Challenges
Ryan Spitler∗, Saeid Zanganeh∗, Tahereh Jafari†,
Nasser Khakpash‡, Mohsen Erfanzadeh§, Jim Q. Ho¶,
and Nastaran Sakhaie||
∗Department of Radiology, Molecular Imaging Program at Stanford,
Stanford University, CA, USA
†Institute of Materials Science, University of Connecticut,
97 North Eagleville Road, Storrs, CT, USA
‡Department of Chemical and Biomolecular Engineering, University of
Connecticut, 191 Auditorium Road, Storrs, CT, USA
§Department of Biomedical Engineering, University of Connecticut,
Storrs, CT, USA
¶Middlebury College, Middlebury, VT, USA
||Department of Nanotechnology, Faculty of Pharmacy,
Tehran University of Medical Sciences, Tehran, Iran
Abbreviations
| I. | Introduction |
| II. | Liposomal and Targeted Drug Delivery System A.Evolution of liposomal drug targeting B.Liposome classification C.Types of liposomal drug delivery platforms D.Passive targeting 1.Limitations of passive targeting E.Active targeting F.Challenges in liposomal drug delivery |
| III. | Transdermal Drug Delivery A.Passive transdermal drug delivery B.Active transdermal drug delivery C.Transdermal delivery system design D.Transdermal drug delivery systems technologies 1.Polymer membrane partition-controlled TDD systems 2.Polymer matrix diffusion-controlled TDD systems 3.Drug reservoir gradient-controlled TDD systems 4.Micro-reservoir dissolution-controlled TDD systems E.Transdermal drug delivery challenges |
| IV. | Microemulsion Drug Delivery System A.Microemulsion in pharmaceuticals 1.Transdermal route 2.Nasal route 3.Oral route 4.Parenteral route 5.Ocular delivery 6.Tumor targeting 7.Cancer treatment B.Microemulsion challenges |
| V. | Nanotechnology for Drug Delivery Systems A.Polymers in drug delivery 1.Liposomes 2.Micelles 3.Dendrimers 4.Nanospheres and nanocapsules B.Inorganic nanoparticles 1.SPIONs C.Nanotechnology challenges |
| VI. | Concluding Remarks |
References
Abbreviations
CPP | cell penetrating peptides |
CRT | controlled-release technology |
DSPE | distearoylphosphatidylethanolamine |
DDS | drug delivery systems |
EPR | enhanced permeability and retention |
EMEA | European Medicine Evaluation Agency |
FDA | Food and Drug Administration |
LDL | low density lipoprotein |
LUV | large unilamellar liposomes |
mPEG | methoxy polyethylene glycol |
MNs | micro needles |
NPs | nanoparticles |
PEG-PE | poly(ethylene) glycol-phosphatidylethanolamine |
PEG | polyethylene glycol |
RES | reticuloendothelial system |
SPIONs | superparamagnetic iron oxide |
TDDS | targeted drug delivery system |
mAb | monoclonal antibodies |
TATp | transcription protein |
TDDS | transdermal drug delivery systems |
I.Introduction
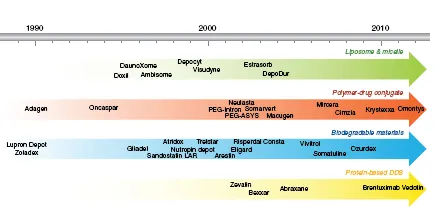
Drug delivery systems (DDS) have the capability to improve the administration and efficacy of treatment for pharmaceutical compounds such as antibodies, peptides, vaccines, drugs, enzymes, and many others. DDS have the potential to improve the quality of life and longevity of millions of patients. The ability to create controlled release formulations of existing drugs has changed the economics of drug development. Thereby, DDS can improve performance and efficacy, as well as extend the patent life of an existing compound by creating a new product. New DDS are less costly and can be developed much faster in comparison to (approximately $20–50 million and 3–4 years) new drugs (approximately $500 million in 10 years).1 The US market for DDS has demonstrated substantial growth from $75 million in 2001 to as much as $121 billion in 2010.2 In 1990, Liposomal amphotericin B was the first Food and Drug Administration (FDA) approved DDS. There are currently more than 10 DDS which are commercially available for the treatment of a broad range of diseases from cancer, fungal infections, and muscular degeneration.3 Figure 1 shows the timeline of FDA-approved DDS in the market.4
However, a significant limitation for any drug therapy is the inherent cytotoxicity. Upon administration, many compounds will circulate systemically and may cause substantial toxicity. The possible occurrence of these harmful effects, post treatment, can significantly limit the clinical application of many new compounds. In many cases controlled delivery by DDS mitigates many of these drawbacks by improving the following properties: bioavailability, preventing premature degradation, enhancing uptake, maintaining and controlling the drug release rate, and reducing adverse effects by using highly specific targeting approaches.
Figure 1. Timeline showing FDA approved DDS in the market. Taken from Ref. [4] with permission of Elsevier.
One example of a targeting approach is the use of site-specific pharmaceutical nanocarriers. The drug targeting of these nanocarriers has been extensively studied and they possess the following advantages: altered drug distribution dynamics, increased delivery to target sites, simplified drug administration methods, improved efficacy and reduced cost.5 One example are liposomes as they are well-characterized nanocarriers and they will be discussed in detail in the following DDS sections.
Nanocarriers have been used in numerous animal and human investigations and have utilized alternate routes of administration for analgesic and anesthetic, using delivery through the skin, buccal, and nasal mucous membranes. In work to improve these delivery methods, other compounds have lead to an abundance of new devices, concepts and techniques that have collectively been termed controlled-release technology (CRT). Some examples of CRT include transdermal controlled release delivery systems, ml6 nasal and buccal aerosol sprays, drug-impregnated lozenges, encapsulated cells, oral soft gels, drugs administered through the skin and various programmable, and implanted drug-delivery devices. There are numerous motivating factors driving the development of these new devices, concepts and techniques including unmet clinical need, sustained efficacy of treatment and the demand for multiple forms of a given deliverable drug.6 Some recent DDS platforms have implemented oral, injectable systems, transdermal,7 ocular8 and nasal routes9 that are entering clinical trials and with some already on the market. In order to achieve successful clinical translation, DDS must be safe, perform the intended therapeutic function, offer a convenient administration method and must be scalable.
Of the many options for controlled release DDS, transdermal delivery has received much attention due to a number of distinct advantages, which will be discussed later in this chapter. One example of a popular drug delivery platform is the microemulsion. This drug delivery system is highly attractive for pharmaceutical formulators due to the relatively simple formulation process. Microemulsions can also be easily utilized for developing novel carriers for phytoactives and extracts.10 In the following sections, microemulsion DDS will be discussed in more detail.
Nanotechnology has interfaced very closely in the past decade with drug delivery.4,11,12 The size and shape of nanoparticles (NPs) are a critical component...