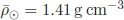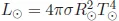![]()
Chapter 1
Observable Properties of Stars
“A star is basically a pretty simple structure” announced Fred Hoyle at a colloquium in the Cambridge Observatory’s Library in 1954. From the audience Professor Redman retorted, “You’d look pretty simple, Fred, at ten parsecs.”1 This was at the dawn of the modern age of stellar astrophysics. The basic physics had been identified and the equations assembled. It remained to solve them in progressively more detail and so model the stars in all their glory, a process that continues apace. Today’s instruments and observing techniques mean that stars do not look quite so simple at ten parsecs any more, our understanding of the details of the physical processes has improved enormously and we have identified some important complications, particularly in the fluid dynamics of convection and mixing. Nevertheless stars, in their interiors, are indeed pretty simple structures. Not far below their visible surfaces, temperatures are so hot that the complexities of chemistry are not a burden. Even in the atmospheres much of the chemistry appears to be relatively simple and biology remains out of reach. If we understand physics we can understand stars. One of the beauties of stars is that they touch upon every aspect of physics from the quantum mechanics of electron degeneracy and nuclear fusion, the general relativity of the neutron stars and black holes, thermodynamics and electrodynamics, fluid dynamics and magnetohydrodynamics through to solid state crystalline structures of cold white dwarfs. In the coming chapters the necessary physics is described, with some mathematical detail, to the extent that the reader can grasp the essentials and understand the details to the level required to comprehend the current literature and to proceed to push forward the frontiers of our understanding of stars.
First we shall briefly review what we can observe and measure for real stars and so learn what our models must explain. Life on Earth depends on our closest star, the Sun, but it is sometimes easy to overlook its stellar nature just because of its very proximity. It is only because the Earth orbits at a distance that allows our carbon-based life to evolve and survive, sustained by the energy the Sun radiates, that we can contemplate the stars at all. Its proximity allows us to study the Sun in quite intimate detail compared to our distant, apparently point-like, neighbours. Hence we naturally use the Sun as a standard with which we compare all other stars. From even a minimal study we can deduce three important facts about stars: they are rather luminous, hot and apparently spherical.
Early scientific study of the Sun can be traced back to observations of sunspots by Chinese astronomers as early as 206 BC. Similar observations were also undertaken by the medieval Andalusian polymath Averës in the twelfth century. However it was not until the invention of the telescope in the seventeenth century that detailed observations could be undertaken by the likes of Thomas Harriot and Galileo Galilei. By following the motion of spots on the Sun’s surface Galileo deduced that it rotates with a period of about a month. However this rotation is not the uniform rotation of a solid body. The period varies from about 35 d at the poles to 24.5 d at the equator. This corresponds to an equatorial surface velocity of 1.7kms−1. Today we know that this, though typical of stars like the Sun, is rather slow compared with many more massive stars.
In the same century Johannes Kepler deduced his laws of planetary motion from meticulous analysis of observations made by Tycho Brahe. Soon after, Sir Isaac Newton’s laws of motion and gravity made it possible to deduce the relative masses of the Sun and the planets from the periods of their satellites and trigonometric estimates of distances. Though such estimates of the distance to the Sun from Earth have been made since ancient times an accurate absolute scale was missing until the transits of Venus in 1761 and 1769 were observed from distant points on the surface of the Earth in a major international collaboration. This not only gives the mean distance from the Earth to the Sun, the astronomical unit (a⊕ = 1AU), but also the solar radius and an estimate of the Sun’s absolute luminosity from measurements of its energy flux through a unit area at the Earth, the Solar irradiance. The absolute masses of the solar system bodies required the further measurement of Newton’s gravitational constant which did not come until the late eighteenth century when Henry Cavendish completed John Michell’s2 proposed measurement of the gravitational force between two masses on the Earth.
These fundamental parameters of the Sun are measured more and more precisely as techniques for their measurement change and improve. In summary3
•the solar mass, M⊙ = 1.9885 × 1033 g = 1.9885 × 1030 kg,
•the solar radius, R⊙ = 6.957 × 1010 cm = 6.957 × 108 m,
•the solar luminosity, L⊙ = 3.828 × 1033 ergs−1 = 3.828 × 1026 W and
•the astronomical unit, 1 AU = 1.496 × 1013 cm = 215 R⊙,
from which we can derive
•
the mean density of the Sun,
and
•the effective temperature, T⊙ = 5 772K,
where the effective temperature of the photosphere of the Sun is defined to be the temperature of a black body of the same radius and luminosity so that
.
Accurate estimates of the age of the Sun came much later. It must be older than the solar system and the Earth itself but it was not until the geological record of the Earth was analysed carefully in the early nineteenth century that an age in excess of ten million years was even contemplated. Today the best estimates of the age of the solar system are based on the radioactive decay of long lived isotopes that were trapped in rocks on the Earth and meteorites when the solar system formed. This method was originally employed by Sir Ernest Rutherford (1929) but did not become reliable until the 1950s. The latest results compare the decays of 238U and 235U to 206Pb and 207Pb in meteoritic inclusions which we believe formed earlier in the Sun’s protoplanetary disc. The measurement errors of a few million years are somewhat smaller than the systematic error of about 108 yr it probably took the Sun to form, hence we can be reasonably certain that
•the age of the Sun, t⊙ ≈ 4.6 × 109 yr.
Since Joseph Fraunhofer invented the spectroscope in the early nineteenth century, much has been learnt from analysing the solar spectrum and the spectra of other stars. Most important is the composition of the solar surface. In Chapter 5 we examine how the photosphere of a star emits light in a black-body spectrum and how this is modulated by atomic absorptions further out in the star’s atmosphere (see also Sec. 1.3.5). The shape of the absorption lines gives a plethora of information including the abundances of the absorbing elements, the pressure at which the line forms, any systemic velocity, the broadening caused by the star’s rotation as well as a measure of any magnetic field strong enough to induce Zeeman splitting of the lines.
In 1925 Cecilia Payne (1925) (later Payne-Gaposchkin) used Meghnad Saha (1921)’s ionisation theory (Sec. 3.6) applied to spectra to determine abundances of various elements in stellar atmospheres. Though she found many elemental abundance ratios to be consistent with terrestrial ratios she unexpectedly found the lightest elements, hydrogen and helium, to dominate. This was the first indication that the composition of stars is very different to that of the Earth. In 1926 when Eddington was postulating hydrogen fusion as the source of the Sun’s luminosity he was still reluctant to admit a hydrogen abundance above the 7% he believed necessary to power the Sun for 1010 yr. The dominance of hydrogen and helium was only confirmed when accepted for the Sun by Henry Norris Russell (1929). Helium emission lines can be seen in the solar chromosphere during eclipse, and indeed the new element helium was first identified this way by Norman Pogson and named in 1868 by Sir Norman Lockyer. However it is not possible to make an absolute determination of the abundance of the Sun’s hydrogen and helium spectroscopically because the electrons in helium are too tightly bound at the temperature of its photosphere. Measurements of composition are limited to elements more massive than helium, known collectively as metals, for which abundances relative to hydrogen can be found from spectra. Though the Sun’s helium abundance can be estimated with other techniques, such as helioseismology which is able to probe how helium ionises with depth in a way that depends on its abundance, direct measurements of the solar wind or chromospheric observations, it is not well constrained and is usually fixed to ensure that solar models have the correct luminosity (Sec. 8.4). We shall use canonical abundances by mass for hydrogen X = 0.7, helium Y = 0.28 and metals, the metallicity Z = 0.02 so that Z/X = 0.029. Recent analysis of the solar spectrum, taking into account three-dimensional modelling of convective motions apparent in the solar granulation, suggests that this is too high for the solar photosphere where something as low as Z/X = 0.018 may be more appropriate. However helioseismological measurements remain more consistent with higher metallicity. As we write, this remains an active area of research in which a resolution is still awaited.
The relative abundances of the metals are better constrained both by the solar spectrum and by, mostly consistent, measurements on the Earth and within the solar system. However it is particularly difficult to measure abundances of all the noble gases, He, ...