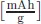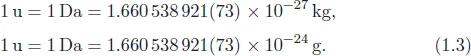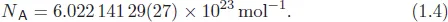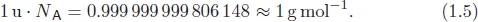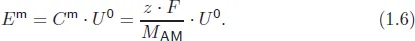![]()
Chapter 1
Fundamental Aspects of Achievable Energy Densities in Electrochemical Cells
Kai Peter Birke and Desirée Nadine Schweitzer
Currently, lithium-ion batteries are the most common energy sources for today’s portable devices like laptops and smartphones, but their fields of application extend to more demanding sectors such as the automotive and smart grid sectors. The concept of electrochemically rechargeable energy storage is still quite complex, though many simplifications of cell chemistry and suppression of undesirable side reactions have already taken place after its transition from lead acid to Li-ion.
Thus, the improvement of future secondary battery systems seems, at first glance, to be down to its choice of basic cell elements, especially active materials that contain energy.
In this regard the following three main cell characteristics have to be considered: (1) the specific capacity, (2) the specific energy density and (3) the volumetric energy density. However, one important question remains: will the most promising active materials result in the most appropriate battery? In this chapter, a suitable method to collect the best basic cell active materials for a battery will be shown.
The specific capacity, typically provided as [mAh/g] or [Ahkg−1] of an element, shows how much electricity can be stored in one gram of this element. This depends on the number of electrons z that can be transferred in the electrochemical redox reaction [1]. The electrical charge per mole of electrons is defined as the Faraday constant F, which gives, if multiplied with the number of electrons z, the molar capacity:
The value of the Faraday constant is about 96 485 C mol−1. This molar capacity for the given electrochemical redox reaction as a function of the molar mass of the element and the number of transferred electrons (this is the redox-reaction specific part) results in the specific capacity of the single element or chemical compound, as shown in Eq. (1.2). To calculate the specific capacity of a single element, it is necessary to take the ideal assumption that all of the material participates in the electrochemical redox reaction. The value of the molar mass of each element MAM (AM = Active material) can be easily derived from the periodic table [2].
As 1 Coulomb = 1 Ampere × 1 second, it is recommended to divide Eq. (1.2) by the factor 3.6 to obtain the specific capacity
Cm in the appropriate unit
.
The atomic mass is the mass of an atom, which can be expressed in kilograms [kg] (See Eq. (1.3)). Often, the atomic mass unit [u] or Dalton [Da] is used for calculation. The atomic mass is the 12th part of the mass of a carbon isotope 12C, which corresponds in close approximation to the mass of one hydrogen atom, so one can say the weight of a hydrogen atom is 1 u.
Equation (1.4) shows the Avogadro constant NA, which is simply the number of particles in one mole.
Multiplying 1 u with the Avogadro constant delivers a very good approximation of 1 g mol−1. Since the weight of one hydrogen atom can be assumed to be 1 u, Eq. (1.5) shows that the atomic weight of an element can be easily derived by taking the same absolute value.
For example, the lithium element, which has 6.94 u, and by approximation, also 6.94 g mol−1.
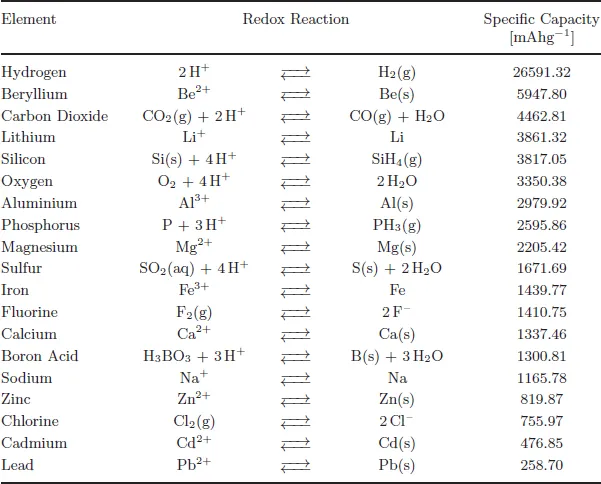
Table 1.1Specific capacity of selected elements and corresponding redox reaction [1].
Table 1.1 summarizes the specific capacity of common elements — some already in use in commercialized batteries, while others are discussed as potential promising candidates or as examples why their high capacities cannot be transferred to modern batteries. The full list of all these elements and their positions in the electrochemical series is collected in Appendix A.
Because most electrochemical redox reactions that can be efficiently employed in electrochemical cells to store energy need only the transfer of one or two electrons, high values of specific capacities can be consequently found in the upper left and right rows of the periodic table. Therefore, as a rule of thumb, the specific capacity depends mostly on the molar mass of an element. The lightest elements, with low molar masses are located in the first rows of the periodic table. For example, the electrochemical redox reaction of lithium, which generally transfers one electron only, results in a high specific capacity value due to its low molar mass. However, the specific capacity of silicon results in more or less the same value though its molar mass is four times higher than the molar mass of lithium because it uses four electrons in the redox reaction process. The same holds for aluminium with a three-electron transfer. In this regard aluminium and silicon are exceptions since no other elements can compensate their higher molar mass by electron transfer in such an efficient way. Lithium and silicon have very high specific capacities, so both could be, at this point, potential electrode materials for secondary battery systems.
Besides capacity, the voltage, as compared to hydrogen, strongly affects the energy density in the materials vying to be the best choice for cell components in a secondary battery system. The energy density represents how much energy can be stored in the cell and sums up the absolute amount of the voltages of the two single electrodes, as compared to a reference, usually hydrogen or in case of Li-based cells, Li. This means that the voltage and the specific electrode capacities have to be always considered.
But, is it also feasible that the gravimetric energy density Em of an element or compound will be able to provide a deeper insight than its specific capacity? The gravimetric energy density of an element shows how much energy per kilogram or gram can be stored, while its volumetric energy density shows how much energy per litre can be stored. In this regard these are the most important values to differentiate potential candidates for high-energy cells. To calculate the gravimetric energy density Em of a single element or compound, the specific capacity Cm is multiplied with the electrochemical series voltage U0, as shown in Eq. (1.6).
In the following, the values for the electrochemical series voltages are based on the hydrogen level. This means that the electrochemical redox reaction of hydrogen has the voltage of U0 = 0 V. Thus, one has to be careful with this value. However, for the choice of battery materials it makes some sense since hydrogen represents the middle (~3V) of the reduction direction, and oxidation can be achieved. This also means that one single electrochemical cell can never exceed 5.9V, which is the combination voltage of lithium and fluorine!
The series voltages of many relevant elements are listed in Appendix B. Since there are positive and negative values for the series voltages, it formally results in positive and negative gravimetric energy densities per element using hydrogen as a reference.
The values of the gravimetric energy densities of each element can thus be put into a reliable order employing the electro...