For the 6th Edition of this highly regarded textbook devoted to lipids, the title has been modified from Lipid Biochemistry to Lipids to acknowledge the coming together of biological and medical sciences, the increasingly blurred boundaries between them and the growing importance of lipids in diverse aspects of science and technology. The principal aims of this new edition - to inform students and researchers about lipids, to assist teachers and encourage further research – have not changed since previous editions.
Significant advances in lipid science have demanded yet another extensive rewriting for this edition, with the addition of two new authors, to cover new knowledge of genes coding for proteins involved in lipid metabolism, the many lipids involved in cell signalling, the roles of lipids in health and disease and new developments in biotechnology in support of agriculture and industry.
An introductory chapter summarizes the types of lipids covered and their identification and provides a guide to the contents. Chapters contain boxes illustrating special topics, key point summaries and suggested further reading.
Lipids: Sixth Edition provides a huge wealth of information for upper-level students of biological and clinical sciences, food science and nutrition, and for professionals working in academic and industrial research. Libraries in all universities and research establishments where biological, medical and food and nutritional sciences are studied and taught should have copies of this excellent and comprehensive new edition on their shelves.

eBook - ePub
Lipids
Biochemistry, Biotechnology and Health
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Lipids
Biochemistry, Biotechnology and Health
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Chapter 1
Lipids: Definitions, Naming, Methods and a Guide to the Contents of this Book
1.1 Introduction
Lipids occur throughout the living world in microorganisms, fungi, higher plants and animals. They occur in all cell types and contribute to cellular structure, provide energy stores and participate in many biological processes, ranging from transcription of genes to regulation of vital metabolic pathways and physiological responses. In this book, they will be described mainly in terms of their functions, although on occasion it will be convenient, even necessary, to deal with lipid classes based on their chemical structures and properties. In the concluding section of this chapter, we provide a ‘roadmap’ to help students find their way around the book, so as to make best use of it.
1.2 Definitions
Lipids are defined on the basis of their solubility properties, not primarily their chemical structure.
The word ‘lipid’ is used by chemists to denote a chemically heterogeneous group of substances having in common the property of insolubility in water, but solubility in nonaqueous solvents such as chloroform, hydrocarbons or alcohols. The class of natural substances called ‘lipids’ thus contrasts with proteins, carbohydrates and nucleic acids, which are chemically well defined.
The terms ‘fat’ and ‘lipid’ are often used interchangeably. The term fat is more familiar to the layman for substances that are clearly fatty in nature, greasy in texture and immiscible with water. Familiar examples are butter and the fatty parts of meats. Fats are generally solid in texture, as distinct from oils which are liquid at ambient temperatures. Natural fats and oils are composed predominantly of esters of the three-carbon alcohol glycerol with fatty acids, often referred to as ‘acyl lipids’ (or more generally, ‘complex lipids’). These are called triacylglycerols (TAG, see Section 2.2: often called ‘triglycerides’ in older literature) and are chemically quite distinct from the oils used in the petroleum industry, which are generally hydrocarbons. Alternatively, in many glycerol-based lipids, one of the glycerol hydroxyl groups is esterified with phosphorus and other groups (phospholipids, see Sections 2.3.2.1 & 2.3.2.2) or sugars (glycolipids, see Section 2.3.2.3). Yet other lipids are based on sphingosine (an 18-carbon amino-alcohol with an unsaturated carbon chain, or its derivatives) rather than glycerol, many of which also contain sugars (see Section 2.3.3), while others (isoprenoids, steroids and hopanoids, see Section 2.3.4) are based on the five-carbon hydrocarbon isoprene.
Chapter 2 deals mainly with lipid structures, Chapters 3 and 4 with biochemistry and Chapter 5 with lipids in cellular membranes. Aspects of the biology and health implications of these lipids are discussed in parts of Chapters 6–10 and their biotechnology in Chapter 11. The term ‘lipid’ to the chemist thus embraces a huge and chemically diverse range of fatty substances, which are described in this book.
1.3 Structural Chemistry and Nomenclature
1.3.1 Nomenclature, General
Naming systems are complex and have to be learned. The naming of lipids often poses problems. When the subject was in its infancy, research workers gave names to substances that they had newly discovered. Often, these substances would turn out to be impure mixtures but as the chemical structures of individual lipids became established, rather more systematic naming systems came into being and are still evolving. Later, these were further formalized under naming conventions laid down by the International Union of Pure and Applied Chemistry (IUPAC) and the International Union of Biochemistry (IUB). Thus, the term ‘triacylglycerols’ (TAGs – see Index – the main constituents of most fats and oils) is now preferred to ‘triglyceride’ but, as the latter is still frequently used especially by nutritionists and clinicians, you will need to learn both. Likewise, outdated names for phospholipids (major components of many biomembranes), for example ‘lecithin’, for phosphatidylcholine (PtdCho) and ‘cephalin’, for an ill-defined mixture of phosphatidylethanolamine (PtdEtn) and phosphatidylserine (PtdSer) will be mostly avoided in this book, but you should be aware of their existence in older literature. Further reference to lipid naming and structures will be given in appropriate chapters. A routine system for abbreviation of these cumbersome phospholipid names is given below.
1.3.2 Nomenclature, Fatty Acids
The very complex naming of the fatty acids (FAs) is discussed in more detail in Chapter 2, where their structures are described. Giving the full names and numbering of FAs (and complex lipids) at each mention can be extremely cumbersome. Therefore a ‘shorthand’ system has been devised and used extensively in this book and will be described fully in Section 2.1, Box 2.1. This describes the official system for naming and numbering FAs according to the IUPAC/IUB, which we shall use routinely. An old system used Greek letters to identify carbon atoms in relation to the carboxyl carbon as C1. Thus, C2 was the α-carbon, C3 the β-carbon and so on, ending with the ω-carbon as the last in the chain, furthest from the carboxyl carbon. Remnants of this system still survive and will be noted as they arise. Thus, we shall use ‘3-hydroxybutyrate’, not ‘β-hydroxy-butyrate’ etc.
While on the subject of chain length, it is common to classify FAs into groups according to their range of chain lengths. There is no standard definition of these groups but we shall use the following definitions in this book: short-chain fatty acids, 2C–10C; medium-chain, 12C–14C; long-chain, 16C–18C; very long-chain >18C. Alternative definitions may be used by other authors.
1.3.3 Isomerism in Unsaturated Fatty Acids
An important aspect of unsaturated fatty acids (UFA) is the opportunity for isomerism, which may be either positional or geometric. Positional isomers occur when double bonds are located at different positions in the carbon chain. Thus, for example, a 16C monounsaturated (sometimes called monoenoic, see below) fatty acid (MUFA) may have positional isomeric forms with double bonds at C7-8 or C9-10, sometimes written Δ7 or Δ9 (see Box 2.1). (The position of unsaturation is numbered with reference to the first of the pair of carbon atoms between which the double bond occurs, counting from the carboxyl carbon.) Two positional isomers of an 18C diunsaturated acid are illustrated in Fig. 1.1(c,d). Geometric isomerism refers to the possibility that the configuration at the double bond can be cis or trans. (Although the convention Z/E is now preferred by chemists instead of cis/trans, we shall use the more traditional and more common cis/trans nomenclature throughout this book.) In the cis form, the two hydrogen substituents are on the same side of the molecule, while in the trans form they are on opposite sides (Fig. 1.1a,b). Cis and trans will be routinely abbreviated to c,t (see Box 2.1).
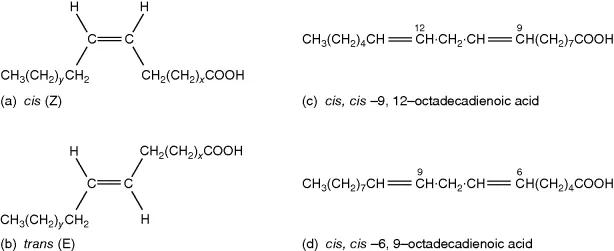
Fig. 1.1 Isomerism in fatty acids. (a) cis-double bond; (b) a trans-double bond; (c) c,c-9,12-18:2; (d) c,c-6,9-18:2.
1.3.4 Alternative Names
Students also need to be aware that the term ‘ene’ indicates the presence of a double bond in a FA. Consequently, mono-, di-, tri-, poly- (etc.) unsaturated FAs may also be referred to as mono-, di-, tri- or poly- (etc.) enoic FAs (or sometimes mono-, di-, tri- or poly-enes). Although we have normally used ‘unsaturated’ in this book, we may not have been entirely consistent and ‘-enoic’ may sometimes be encountered! Furthermore it is important to note that some terms are used in the popular literature that might be regarded as too unspecific in the research literature. Thus shorthand terms such as ‘saturates’, ‘monounsaturates’, ‘polyunsaturates’ etc. will be avoided in much of this text but, because some chapters deal with matters of more interest to the general public, such as health (Chapter 10) and food science or biotechnology (Chapter 11), we have introduced them where appropriate, for example when discussing such issues as food labelling.
1.3.5 Stereochemistry
Another important feature of biological molecules is their stereochemistry. In lipids based on glycerol, for example, there is an inherent asymmetry at the central carbon atom of glycerol. Thus, chemical synthesis of ph...
Table of contents
- Cover
- Title Page
- Copyright
- Preface
- Acknowledgements
- About the Authors
- About the Companion Website
- Chapter 1: Lipids: Definitions, Naming, Methods and a Guide to the Contents of this Book
- Chapter 2: Important Biological Lipids and their Structures
- Chapter 3: Fatty Acid Metabolism
- Chapter 4: The Metabolism of Complex Lipids
- Chapter 5: Roles of Lipids in Cellular Structures
- Chapter 6: Dietary Lipids and their Biological Roles
- Chapter 7: Lipid Assimilation and Transport
- Chapter 8: Lipids in Transmembrane Signalling and Cell Regulation
- Chapter 9: The Storage of Triacylglycerols in Animals and Plants
- Chapter 10: Lipids in Health and Disease
- Chapter 11: Lipid Technology and Biotechnology
- Index
- End User License Agreement
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Lipids by Michael I. Gurr,John L. Harwood,Keith N. Frayn,Denis J. Murphy,Robert H. Michell in PDF and/or ePUB format, as well as other popular books in Biological Sciences & Biochemistry. We have over 1.5 million books available in our catalogue for you to explore.