![]()
CHAPTER ONE
THE ATMOSPHERE
Quite simply the atmosphere is the air surrounding our planet Earth – mercifully kept in place by the force of gravity.
It is primarily composed of 78% nitrogen, 1% of other gases and, fortunately, 21% oxygen for which we should be truly grateful. Apart from life as we know it being unable to survive without oxygen, combustion would not be possible for engines to function.
As the force of gravity is greater nearest to its source, so the atmosphere is more concentrated near our planet’s surface.
There are four basic factors that affect the weather and flight.
They are:
DENSITY |
– the weight of a given volume of air. |
PRESSURE |
– the weight of a given column of air in the atmosphere. |
TEMPERATURE |
– the warmth of the atmosphere. |
HUMIDITY |
– the moisture content of the atmosphere. |
For occasions when uniformity or a working standard is required, the International Civil Aviation Organisation (ICAO) has agreed on an International Standard Atmosphere (ISA). This lays down standards for density, pressure and temperature based on a defined sea level — more on this later.
You must remember that these standards are but a ‘yardstick’ and they are purely theoretical. Rarely would the conditions in the atmosphere ever coincide with the standards all at the same time.
How these ISA standards play a part in aviation will be made clear as we progress.
![]()
CHAPTER TWO
DENSITY
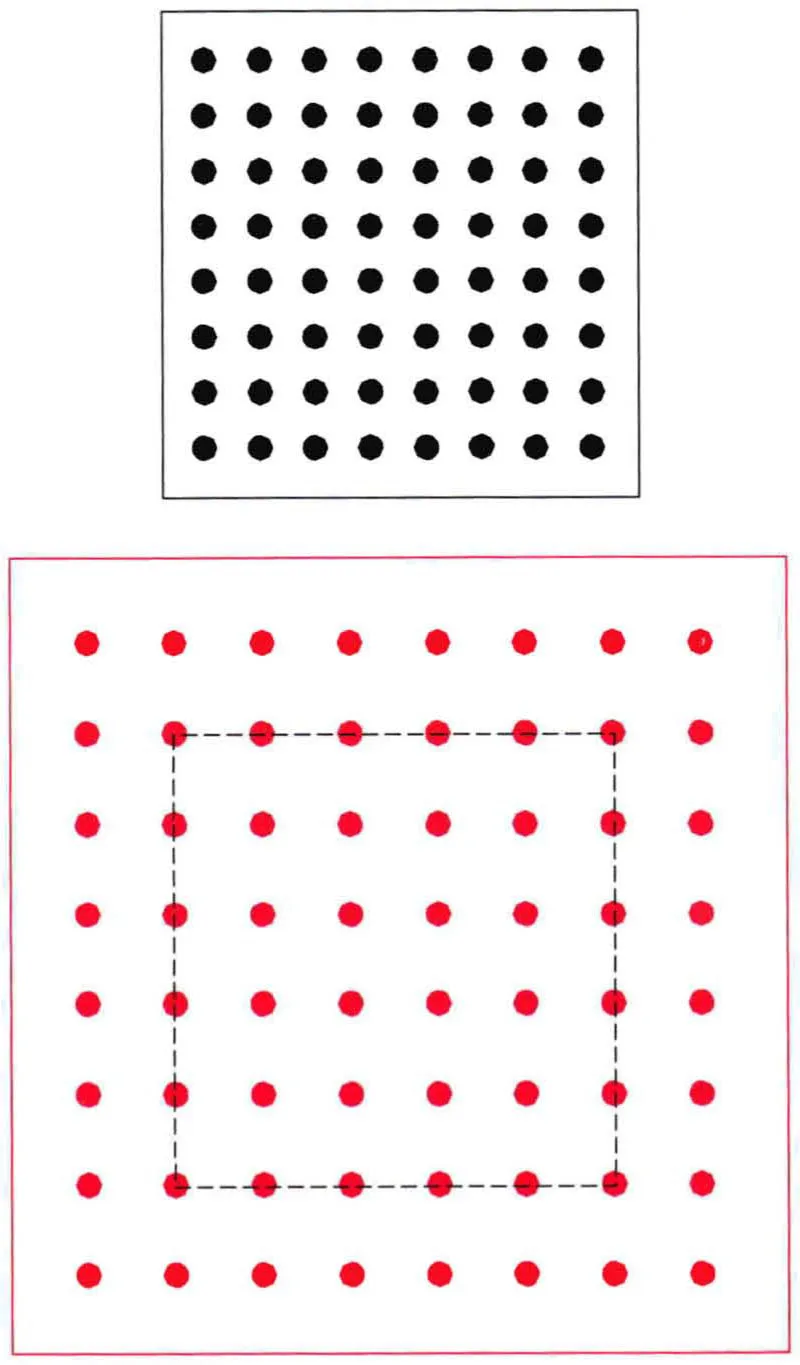
Air has weight, and density is simply the weight of the number of molecules of air present in a given ‘parcel’ or volume of air at a given time. (The word parcel is frequently used in meteorology as no one has yet thought of a better alternative!)
Density is greatest at the surface; it decreases with height as the air thins out until in outer space it is no longer relevant.
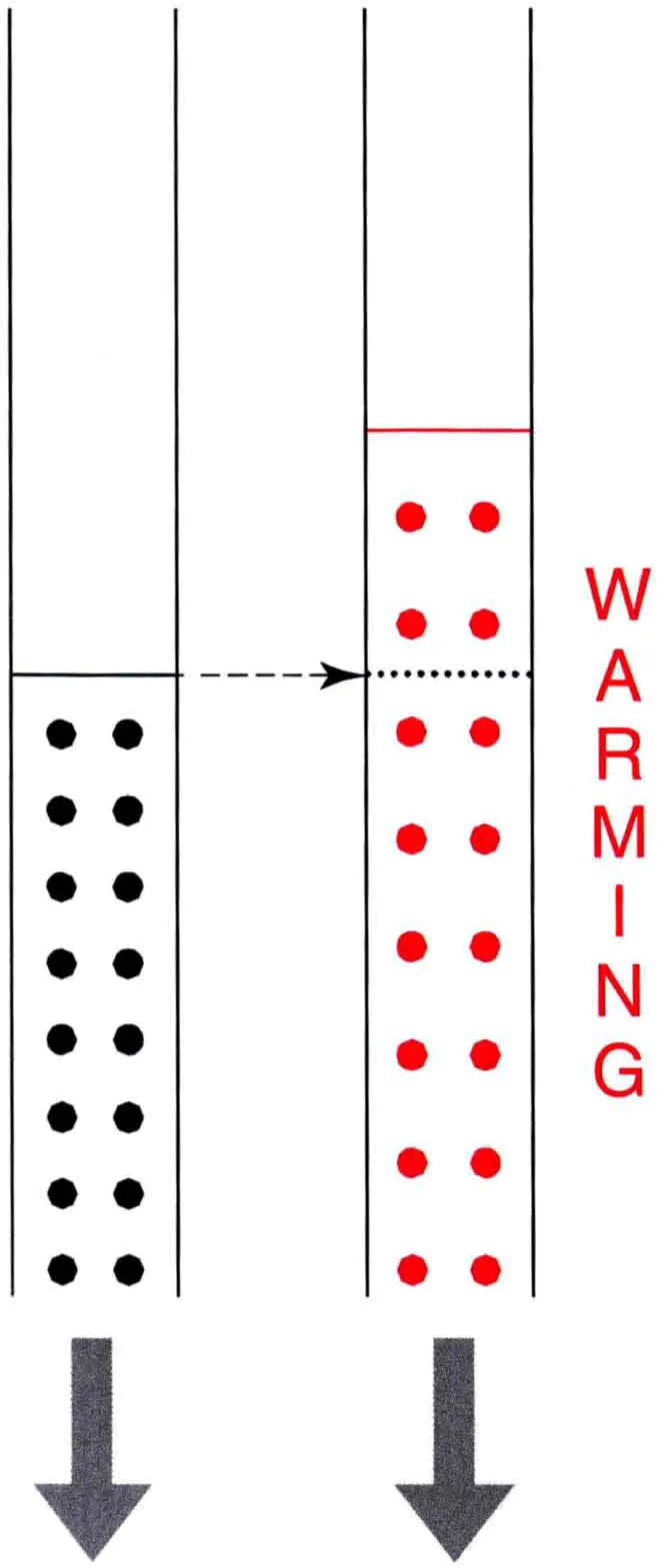
Apart from a decrease with height, density is also affected by warmth. When a parcel of air of a given volume is heated it becomes thinner as the molecules spread out and their number becomes less than in the original parcel.
Conversely, when a volume of air is cooled it becomes denser.
Density also decreases as the moisture content in the air increases, but the effect is relatively minimal.
The ISA standard density at sea level (excepting the Dead Sea!) is 1.225 kilograms per cubic metre (or 1225 grams) or 0.764 lb per cubic foot, where density is defined as 100%. In other words, this would be the defined average weight of all the air molecules in a volume of one cubic metre of air at sea level.
Unlike the other basic factors we shall cover, it cannot be readily measured by instrumentation – its value at a given time requires calculation.
Density plays an important part in an aircraft’s performance; in fact an aircraft’s design limitations are based on the ISA standards. However, as already mentioned, reality rarely matches laid down standards; they are but yardsticks.
The effect of density below the established 100% at 1.225 kg per cubic metre has a definite bearing on an aircraft’s stated ‘book’ performance. This area we shall discuss after you have digested the aspects of pressure, temperature and humidity.
Warmed air expands – notice how the number of molecules in the original volume has decreased
![]()
CHAPTER THREE
PRESSURE
WHAT IT IS
Pressure is the weight of a given column of air in the atmosphere measured at the earth’s surface. It decreases with height until becoming nil when air ceases to exist. You may think that the reference to pressure being weight means it is the same as density — this is not so.
Imagine warming a given parcel of air in a tube. This air would expand and spread itself farther up the tube so that the column in the tube would become greater in length. But take the volume of the original parcel of air in the longer column and you will find it is lighter (less dense) than it was before the warming took place. The same amount of air remains in the column overall so the pressure at the bottom stays the same.
The surface pressure need not decrease with a decrease in density
MEASURING PRESSURE
The unit by which atmospheric pressure is measured in the UK has for many years been the millibar (mb). The ICAO has now agreed to a change of unit to that of the hectoPascal (hPa). The change is rather academic in that 1 mb equals 1 hPa so it is a case of “a rose by any other name”!
However, the millibar will continue to be used in the UK for operational purposes for the foreseeable future; the hectoPascal will be confined only to those occasions when reference is being made to ICAO International Standard Atmosphere as a definition in itself.
In the light of this present situation we will naturally refer to millibars (mbs) in this book with possibly the occasional insertion of hPa as a reminder for the future.
In the United States of America, measurement of pressure is currently in inches (in.Hg) – based on the length of a column of mercury in circumstances about to be described.
The mercury barometer consists of a long thin tube extending upwards from a reservoir of mercury. A tiny hole in the reservoir allows air pressure to enter and exert its influence on the mercury. The above is an example from where the inch is the unit of measurement
Readily recognisable with its clock face and scale marked with a range of choices from ‘Set Fair’ to ‘Stormy’, or simply a plain dial showing measurement only. A perfectly reaso...