1
Expanding Roles and Regulatory Networks of LadS/RetS in Pseudomonas aeruginosa
Chuanmin Zhou, Maryam Dadashi, and Min Wu
CONTENTS
1.1 The Two-Component Systems
1.2 Discovery of RetS and LadS
1.3 Opposing Roles of RetS and LadS
1.4 Function of LadS/RetS Is Dependent on Small RNAs
1.5 Other Hybrid Sensor Kinases in Gac/Rsm Pathway
1.6 Other sRNAs as Regulators
1.6.1 P27 sRNA
1.6.2 PhrS sRNA
1.6.3 RsmV and RsmW sRNAs
1.7 Is RetS a Calcium Sensitive Histidine Kinase?
1.8 Can RetS and LadS Interact with CRISPR-Cas Systems?
1.9 Drug Targets
1.10 Concluding Remarks
References
1.1 The Two-Component Systems
Gram-negative opportunistic pathogen Pseudomonas aeruginosa is a severe host pathogen, found widely in nature, exposing in dynamic environmental conditions. Of note, the two-component system (TCS) is important for sensing those environmental challenges which in turn modulate a number of gene expressions (Stock, Robinson, and Goudreau 2000). Typically, TCS is coupled with a sensor histidine protein kinase and a response regulator protein. Histidine protein kinase is responsible for detecting extracellular signals, regulating the downstream effectors in response to the stimuli through phosphorylated response regulator protein (Stock, Robinson, and Goudreau 2000). To date, over 100 TCS genes have been found in P. aeruginosa (Rodrigue et al. 2000, Stover et al. 2000).
1.2 Discovery of RetS and LadS
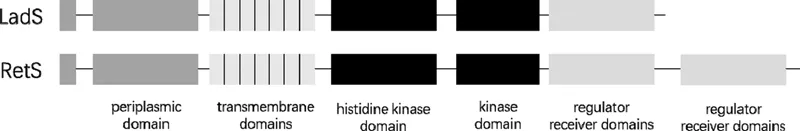
Hybrid sensor kinase RetS (regulator of exopolysaccharide and Type III secretion) was first described in P. aeruginosa in 2004, which encoded 942 amino acids. This protein not only contains N-terminal cleaved signal sequences, a large periplasmic domain, and seven transmembrane domains (associated with environmental signal transduction), but also possesses TCS-like histidine kinase and response regulator domains in tandem, revealing that other TCS regulators may exist (Laskowski, Osborn, and Kazmierczak 2004, Goodman et al. 2004). RetS orthologs were also found in Pseudomonas putida, Pseudomonas fluorescens, Pseudomonas syringae, and Azotobacter vinelandii (Goodman et al. 2004). In 2005, another hybrid sensor kinase named LadS (lost adherence sensor) was noticed in P. aeruginosa PAO1 genome, which showed an opposite role of RetS, promoting biofilm formation and inhibiting T3SS activation (Ventre et al. 2006).
Domain analysis of LadS amino acids showed that LadS contained 795 amino acids with similar domains seen in RetS, including N-terminal cleaved signal sequences, a large periplasmic domain, seven transmembrane domains, as well as a histidine kinase and a response regulator domain (Ventre et al. 2006). These transmembrane domains in LadS and RetS were also observed in a number of other carbohydrate binding proteins (Ventre et al. 2006, Anantharaman and Aravind 2003). In particular, these domains exhibited 35% sequence identity, suggesting that this periplasmic sensor may respond to similar but not 100% identical environmental signals through its unique transmembrane domains (Ventre et al. 2006) (Figures 1.1 and 1.2).
1.3 Opposing Roles of RetS and LadS
RetS was characterized as a global pleiotropic regulatory protein, and the expression levels of almost 400 genes were significantly altered in a retS mutant strain (Goodman et al. 2004). RetS was necessary for the transcription of the T3SS operons under low calcium and host cell contact conditions. Deletion of retS significantly prohibited the activation of T3SS (Laskowski, Osborn, and Kazmierczak 2004). However, no DNA binding motifs were identified in RetS protein domains, indicating that it might modulate T3SS function indirectly (Laskowski, Osborn, and Kazmierczak 2004). The retS deletion strain exhibited robust biofilm formation by promoting the expression of biofilm related genes pel and psl (Goodman et al. 2004). A mutant with a deletion of ladS behaved in an opposite manner (Ventre et al. 2006). Transcriptome analysis of ladS mutant strain compared to WT strain showed that 79 genes were significantly affected, including that pel and psl gene expression were repressed and T3SS were activated in ladS mutant strain (Ventre et al. 2006). Compared to transcriptome analysis in retS mutant strain, 49% were oppositely regulated in ladS mutant strain, indicating that LadS and RetS signaling transduction pathways are antagonistic (Ventre et al. 2006).
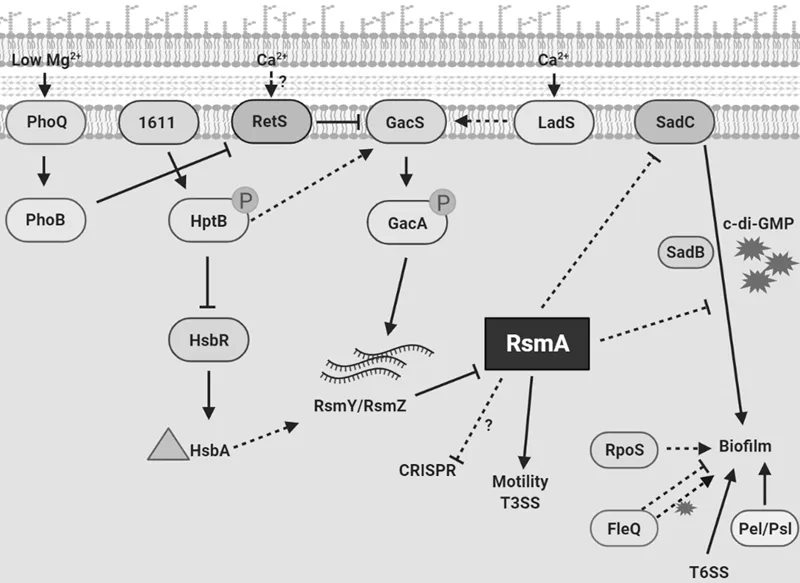
RetS promotes the formation of heterodimers with GacS, reducing the GacS autophosphorylation. Gene screening of transposon insertions showed that RetS may modulate GacS/GacA (Goodman et al. 2004), whereas LadS showed an opposite activity by promoting the activation of GacS/GacA pathway (Ventre et al. 2006). Interestingly, LadS did not interact with GacS and hence may upregulate the GacS/GacA pathway through a phosphor-relay mechanism, resulting in phosphotransfer to the HPT domain of GacS, which in turn promoted chronic infection (Chambonnier et al. 2016). Furthermore, another histidine kinase PA1611 showed a similar role of GacS by interacting with RetS to modulate the GacS/GacA pathway (Kong et al. 2013). The phenotype of retS mutant strain was completely blocked in gacS mutant strain, indicating that RetS works through the GacS/GacA pathway (Ventre et al. 2006). Phosphorylated GacA in turn directly modulated the expression of small noncoding regulatory RNA (sRNA) rsmY/rsmZ. RsmA was a global post-transcriptional regulator, influencing expression of over 500 genes by binding to targeted mRNAs, which was inhibited by sRNA rsmY/rsmZ. Different pathogens including P. aeruginosa, Escherichia coli, Legionella pneumophila, Vibrio cholera, and Salmonella typhimurium, are found expressing rsmA. Finally, deletion of rsmA also showed similar phenotypes to retS deletion mutant strain, exhibiting activation of biofilm and T3SS, which in turn contributes to acute infection (Coggan and Wolfgang 2012).
Although the retS deletion mutant exhibited increased attachment to host cells, it showed less cytotoxicity in eukaryotes and less virulence in a pneumonia mouse model, indicating that the retS mutant strain was unable to respond to environmental signals (Laskowski, Osborn, and Kazmierczak 2004, Goodman et al. 2004). Deletion of ladS showed hyper cytotoxicity compared to the WT strain, and the phenotype of ladS/retS double mutant strain was similar to retS mutant, showing no cytotoxicity, indicating that LadS may function at the upstream of RetS in response to input signals (Ventre et al. 2006) (Figure 1.3).
The stimuli triggering RetS and LadS activity remain largely uncharacterized. Recent research revealed that deletion of ladS causes P. aeruginosa calcium-blind through genetic, biochemical, and proteomic study (Broder, Jaeger, and Jenal 2016). LadS detected calcium, while did not respond to other divalent cations (Mg2+, Fe2+, Zn2+, Mn2+ or Cu2+), through its DISMED2 domain, promoting chronic infection. The presence of an additional helix inhibited the binding of carbohydrate, which in turn promoted the binding of calcium. In addition, deletion of gacA, gacS, and rsmA also showed calcium unresponsive, whereas rsmY or rsmZ single deletion remained calcium sensitive. These studies suggest that calcium signaling plays a key role in host-P. aeruginosa interaction by facilitating acute-to-chronic infection transformation.
1.4 Function of LadS/RetS Is Dependent on Small RNAs
Researchers have studied the mechanism of QS systems for decades. To date, LasI/LasR, RhlI/RhlR QS systems are found important for encoding AHLs, and more than 10% of P. aeruginosa genes are affected by AHLs. It is noticed that production of las and rhl dependent AHLs are positively modulated by two sRNAs, rsmY and rsmZ, and negatively by RsmA. Most QS-dependent genes are repressed by post-transcriptional RsmA effector by bind to its mRNA motif. Considering that LadS/RetS are in the upstream of RsmA, LadS and RetS also showed important roles in influencing QS. Importantly, QS showed important roles associated with acute and chronic infections, indicating that QS is also necessary for acute-to-chronic infection transformation. In addition, RetS contributes in transcriptional repression of sigma factor RpoS, Pel, Psl, and FleQ. FleQ is a repressor of Pel in the absence of c-di-GMP. When c-di-GMP is available, FleQ activates the Pel operon. It was shown that the two-component system PhoQ/B directly interact with RetS. TCS PhoQ/B is a Mg2+ sensing system. When exogenous Mg2+ content is low, the PhoQ/B represses RetS and promotes biofilm formation.
1.5 Other Hybrid Sensor Kinases in Gac/Rsm Pathway
When planktonic cells hit a proper surface, they form biofilms stepwise. Several factors participate in establishment of Pseudomonas biofilms. Below we looked into some role players of biofilm formation. c-di-GMP is a secondary messenger, and its abundance in the cell decides the transition between motility and sessility of the bacterial cells. Biofilms of P. aeruginosa have 75–110 pmol mg−1 c-di-GMP in total cell extracts compared to planktonic cells, which bear merely 30 pmol mg−1 (Basu Roy and Sauer 2014). High levels of c-di-GMP are the hallmark of a biofilm forming lifestyle, which is modulated by diguanylate cyclases like SadC and phosphodiesterases (Merritt et al. 2007).
Recent study indicated that another hybrid sensor kinase, PA1611, modulated genes of acute and chronic infection, which played an important role in downregulation of T3SS and upregulation of biofilm formation (Kong et al. 2013). PA1611 showed similar function to LadS. However, PA1611 did not show a Lads dependent manner. In addition, PA1611 associated with RetS which was similar to GacS showing phosphorelay independent, causing PA1611 shared similar protein domains with GacS (Kong et al. 2013). PA1611 is capable of influencing the Gac/Rsm pathway by promoting the phosphorylation of HptB (Kong et al. 2013). Phosphorylat...