![]()
CHAPTER 1
Micro and Nano-drug Delivery Systems
Delia Mandracchia a and Giuseppe Tripodo*b
a University of Brescia, Department of Molecular and Translational Medicine, Viale Europa 11, 25124 Brescia, Italy
b University of Pavia, Department of Drug Sciences, Viale Taramelli 12, 27100 Pavia, Italy,
*E-mail:
[email protected] Traditional drug formulations deliver the payload by releasing it following relatively simple physical laws, such as drug solubility or controlling its diffusion. The drug cannot always be protected from being degraded by the physiologic environment, or from being released before reaching the site of action. The process of encapsulating an active principle in a micro or nanosystem surrounding the active is itself an enormous scientific advancement in pharmaceutical science. Micro and nanoencapsulation of actives requires a great effort in establishing the best experimental conditions to preserve the drug from premature degradation, to assess the best components of the micro/nanosystem, to provide the most suitable processing conditions and to guarantee the desired therapeutic effect. Last but not least, a possible industrial translation should always be taken into consideration during experimental planning. This chapter aims to give an overview of the preparation and therapeutic applications of micro/nano-drug delivery systems.
1.1 Introduction
Although enormous advancements in pharmaceutical technology have been shown in the last decades, some needs remain unanswered. When considering, e.g., oral drug delivery, significant signs of progress have been made from the simplest acetylsalicylic acid wafers or starch tablets toward the most modern tablets with a controlled drug delivery release. In this way, the first-generation tablets were mostly releasing the loaded drug by following the physicochemical behaviours of the drug, and its bioavailability mainly was depending on them.
Then, in a world-scale attempt to improve the formulation performances, engineers (e.g. industrial machinery like millers, mixers, granulators), physicists (e.g. powder and solution physics) and chemists (e.g. surfactants synthesis, salt form of old drugs) merged their efforts in the newest pharmaceutical technology approach, that exploited these new findings creating modern pharmaceutical science. Where is pharmaceutical technology going today? For sure, it is going in a direction that attempts at targeting the delivered drug to a specific site, hopefully to a specific cell if not a specific genetic sequence. In this way, rethinking a drug delivered not even in a bulk powder but by a micro or nano-cluster would represent a great challenge and opportunity to reach established targets.
There is a dimensional step separating classic formulations (millimetric) from micro and nano, and it is the 1 × 10−3/1 × 10−6 factor that makes the difference between being (much) larger than a cell or much smaller.
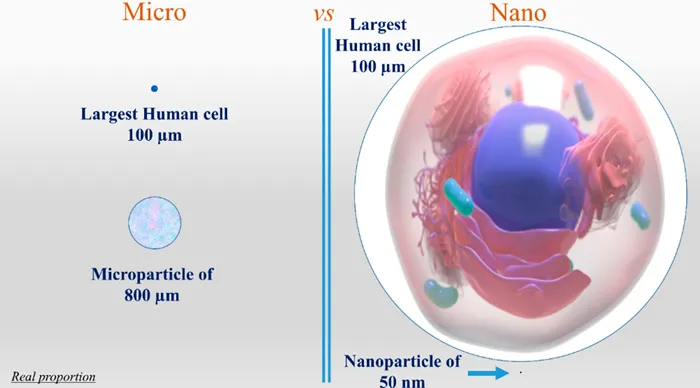
Figure 1.1 gives an overview of how and why one should decide to formulate a drug as micro or nanoparticles. As a general consideration, microparticles will be formulated for the controlled release of actives administered by almost all the routes of administration, with some limitation for the intravenous injection (i.v.) one, depending on size. Nanoparticles, instead, could be delivered by all the known routes of administration with no limits to i.v. Since nanoparticles could be much smaller than human cells, they could be used for intracellular drug delivery and, hopefully, even going deeper to the cellular nucleus. With these premises, microparticles could be most useful as reservoir systems for a controlled drug release, including polypeptide/proteic drugs, while nanoparticles could be most useful for i.v. administration, aiming at a cellular/subcellular target.
Figure 1.1 Micro versus nano: a pictorial representation of the proportions of a human cell and micro or nanoparticles. Cell image reproduced from http://www.medicalgraphics.de/en/free-pictures/organs/cell-white-background.html under the CC BY-ND 4.0 licence, https://creativecommons.org/licenses/by-nd/4.0/.
1.2 Materials Used for Micro or Nano-drug Delivery Systems Preparation
The scientific literature offers a great number of examples where different materials can be used to prepare micro or nanoparticles. Figure 1.2 reports a summary. Essentially, a universal material does not exist. A preliminary classification could be made between inorganic and organic materials, the latter being further divided into natural or synthetic. Concern about the “naturalness” of some materials arises from the obtainment of chemical derivatives of them, which, after that, would not be classified as natural; a known example is cellulose derivatives.
Figure 1.2 Primary materials currently used for the production of micro and nanoparticles.
1.2.1 Inorganic Materials
Iron oxide has received significant attention in the last years for the production of micro and nanosystems for biomedical applications. Iron oxide is considered as a safe material, and its physicochemical behaviours like size, surface characteristics or crystallinity can be easily tuned. 1 The safety of iron oxide particles arises from the fact that iron is an essential element of the human body (e.g. haemoglobin) so entering in the healthy metabolism. The most studied, biocompatible, forms of iron oxide are (i) magnetite (Fe3O4) and (ii) maghemite (γ-Fe2O3). These iron oxides are paramagnetic, a feature that makes them optimal for magnetic resonance imaging. 2,3 However, some toxicity concern arises from the latest experimental evidence, mostly regarding the possibility of reactive oxygen species (ROS) generation and subsequent cellular damage. Therefore, instead of using naked iron oxides, several polymer-coated micro and nanosystems have been proposed. Among those, dextran coating showed good results, such that some of these products were marketed or are in advanced clinical trials. 4,5
Metals like gold and silver have been explored as materials for the production of micro or nanosystems too. Silver is used for its natural antibacterial activity. AgNO3 is commonly used; for example, for the preparation of materials provided with wound healing behaviours. 6 Gold particles saw an increasing interest in their application in different fields of biomedicine. Among the most exciting properties of gold particles, the low toxicity profile should be mentioned, even if their clearance could be difficult. Gold nanoparticles could be obtained in different shapes and sizes, but it has been seen that anisotropic gold particles have peculiar properties even superior to spheroidal particles. 7,8 Gold particles can also be combined with other inorganic materials like silica to form gold nanoshells or prepared as nanocages, nanorods and hollow gold nanospheres. 9,12 One of the keys for the popularity of gold particles is the possibility of their surface functionalization, since the production of gold particles could be performed in the presence of a reducing agent and thiol-bearing molecules which determines the direct functionalization of the particles. 13,14
Silica has been further used for biomedical application. In particular, micro/nanoparticles made from silica found a plethora of applications. 15 Among the silica micro/nanoparticles, mesoporous silica nanoparticles showed great potential. A mesoporous material has pores having a mean diameter of between 2 and 50 nm, according to IUPAC (International Union of Pure and Applied Chemistry). Thus, mesoporous silica micro/nanoparticles show tailorable size between 10 nm to microns and internal pores in the mesoporous range. This allows obtaining particles with tunable physicochemical properties in terms of the loaded drug (molecular dimension of the drug allocated within the pores) and final therapeutic goal, that could be different depending on the size of the particle. Usually, a high amount of drug can be loaded due to the high specific pore volume. Silica particles follow silicon chemistry, which allows the surface derivatisation by reaction with functional silanols. The spherical geometry is not the only one available. Instead, several shapes for mesoporous silica micro/nanoparticles have been developed due to the easy chemical approaches applied for their synthesis. Some examples are hemispheres, dendritic nanoparticles, yolk–shell nanorattles, hollow spheres and Janus Mesoporous Silica nanoparticles (MSNs). 16 Silica-based particles are referred to as biocompatible, even if the real fate of these particles, especially concerning the long term accumulation, is not fully understood. 16
Finally, among the inorganic materials, quantum dots should be mentioned for their broad use in micro/nano-formulated theranostic products. 17
1.2.2 Organic Materials
Organic materials for micro/nanoparticle formulation could be either natural or synthetic. Within this classification, they could be further divided into polymeric/macromolecular (including proteins) or monomeric/oligomeric.
Among monomeric/oligomeric materials, phospholipids could be considered as milestones in the production of micro/nano-drug delivery systems, due to the self-assembling in bilayers that forms liposomes with different sizes, ranging from nanometers to microns. Liposome formulations were among the first nanosystems to be marketed for cancer therapy. 18 The employed phospholipids could be natural or synthetic, and all show a hydrophobic chain and a hydrophilic head. Due to their vesicular structure, liposomes show one or more aqueous compartments and can entrap both lipophilic and hydrophilic drugs. Similar vesicular structures could be formed by “classic” surfactants not bearing phosphate groups like polysorbate; these materials will form the so-called niosomes. Solid lipid nanoparticles are made from lipids that are solid at room temperature and with tailorable melting temperature. The principally used lipids are long-chain alcohols like cetyl alcohol, triglycerides (often homotriglycerides), waxes like cetyl palmitate or bee wax, or sterols. 19 Solid lipids nanoparticles are commonly referred as biocompatible since their main constituents enter in the metabolic pathway of the human body, and thus, are biodegradable too.
Of great importance are also proteic materials like casein, albumin, gelatin and silk proteins such as sericin and fibroin. Casein is the main protein of milk, and it is formed by a protein portion that represents 94% of the components and the remaining 6% so-called colloidal calcium phosphate. 20 It exhibits a Molecular Weight (MW) of about 19–25 KDa and is soluble in water in its sodium caseinate form. It is present in four forms, namely αS1-, αS2-, β-, and κ-casein, and all of them are amphiphilic. Both micro and nanoparticles were prepared with casein. 20,21
Albumin was used to form one of the first nanoparticulate systems reaching the market. 22,23 This marketed product is formed by albumin-bound paclitaxel with a particle size of around 130 nm; these particles injected as such dissolve further in the bloodstream to form smaller 10 nm particles which can spread along the whole body with no capillary occlusion. 24
Another important material for the production of micro/nanoparticles for drug delivery is gelatin. Gelatin is derived from acidic or basic hydrolysis of collagen and is composed of different amino acids (proline, alanine, glycine and others) that confer to the protein a triple helical structure. Gelatin is considered biocompatible and biodegradable. 25,26 To avoid the dissolution of gelatin-based particles at body temperature, the protein can be crosslinked during the formation of the particles by, e.g., glutaraldehyde to control, consequently, the drug release kinetics. 27
A further protein used for micro/nanoparticle preparation is silk and in particular, the two main proteins from which it is composed, fibroin and sericin. Fibroin is present in silk with a percentage between 65 and 85% while sericin is between 15 and 23%, and other components like carbohydrates, salts, wax and other impurities are also present. 28 Fibroin is composed by two leading chains, a heavy (350 KDa) and a light one (25 KDa), and they are linked together by a disulfide bond and further coated by a sericin external layer. Sericin shows a globular structure and has a broad MW ranging 20–400 KDa; it has the function of facilitating the silk spinning from the insects. When mixed to form silk, fibroin and sericin do not show a favourable biocompatibility profile while when employed separately they both are biocompatible. This is why the native silk is “degummed” to separate the two proteins. Both are currently proposed as starting materials to form micro/nanoparticles for biomedical applications. 29,32 The following chapters will provide further details.
As above mentioned, the “universal” material for the preparation of micro/nanoparticles does not exist and, probably, it is why the research is focusing on the use of different polymeric materials that can show different techniques of production, different kinds of entrapped drugs, different fates upon administration and different biodegradability. The main classes of polymer used for micro/nanosystem preparation could be summarised as: (i) polyesters (ii) polysaccharides (iii) polyaminoacids and (iv) polyacrylates. Polyethers like Polyethylene glycol (PEG) or Polypropylene glycol (PPG) and their copolymers are also important. However, often they are not used as such but chemically conjugated to the above polymers. 33,34 Other vinyl polymers like polyvinylpyrrolidone or polyvinyl alcohol are commonly employed as adjuvants during the production of colloidal systems.
Polyesters are milestones in micro and nanoparticles production; they are among the best choices when tailorable mechanical properties, thermal properties, degradation time or crystallinity are required. Al...