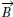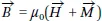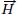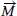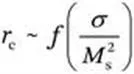a Kansas State University, Department of Chemistry, 213 CBC Building, Manhattan, KS 66506-0401, USA
*E-mail:
[email protected] 1.1 Introduction
Magnetic materials have fascinated people ever since the development of the first compass in 200–300 BC. In a compass, the earth magnetic field interacts with the lodestone providing information about the direction of the magnetic north pole. This piece of information was crucial to establish a universal navigation system leading to close interaction of various cultures and economies throughout this planet. The magnetic compass has gone through several development stages, but the basic idea remains in today’s navigational devices. In nature one finds examples of animals utilizing magnetic nanomaterials for navigation even in the absence of awareness of magnetic fields and magnetic materials. The sensing of the Earth’s magnetic field, called magnetoreception, is present in bacteria, arthropods, mollusks, and even in vertebrates. For instance, Magnetotactic bacteria1 contain several small magnetite crystals that act together as a compass needle to orient these bacteria along the geomagnetic field lines. The small magnetite crystals are created via very well controlled biomineralization processes2 to produce single domain magnetite crystals analogous to the artificial colloidal synthesis of magnetic nanomaterials. Humans are thought to be unaware of the Earth’s magnetic field, however there are molecules such as cryptochrome that, principally, could serve this purpose. From a thermodynamic perspective, magnetic materials can convert the external energy of a magnetic field into heat or energy/work. The type of interaction depends on the nature of the magnetic field, such as homogeneous, inhomogeneous, static or time dependent (dynamic) magnetic fields. These possible interactions between various types of magnetic fields and materials are listed in Table 1.1 adapted from ref. 3. As one can see there is a wide range of effects on materials in general from the different types of magnetic fields. In this chapter, we will review some basic information about magnetic materials with a focus on colloidal magnetic nanoparticles. We will also focus on how colloidal magnetic nanostructures can produce heat when placed in alternating magnetic fields and how these particles can be used to produce mechanical action, e.g. translational motion in the presence of inhomogeneous magnetic fields, specifically the generation of sound waves and their potential applications.
Table 1.1 The impact of various magnetic fields on materials
| Static magnetic fields | Dynamic magnetic fields |
| Homogeneous | Inhomogeneous | Rotating | Oscillating |
| Ferromagnetic materials | Alignment and steering | Alignment and propulsion | Rotational movement | Heat and motion |
| Paramagnetic materials | Alignment and steering | Alignment and propulsion, magnetic trapping | Rotational movement of chains of paramagnetic particles | Induce beating pattern in paramagnetic bead chains |
| Manipulate self-assembly of paramagnetic beads |
| Diamagnetic materials | Alignment and deformation | Magnetic levitation and separation, magnetic trapping | Alignment along multiple axis | No known examples so far |
1.2 Magnetism in Nanoparticles
Magnetic nanoparticles are a group of engineered particles (typically smaller than 150 nm) that can be controlled under the influence of an external magnetic field.
4 In these particles, the magnetic field
is produced by magnetic materials or by free electric currents:
µ0 represents the magnetic permeability of a vacuum and the contribution of
arises from the generation of a free current while the magnetization
can be defined as the induced magnetic dipole moments in a magnetic material. The factors that influence the magnetic properties of nanoparticles include their chemical composition, crystal lattice, particle shape and size, morphology and finally the manner in which the magnetic nanoparticles interact with adjacent particles or with the surrounding matrix.
5 When the size of the nanoparticles decreases, the surface-to-volume ratio increases. Exploring the properties of the large surface-to-volume ratio of the nanoparticles is of critical importance and has led to novel physical, chemical, and mechanical property discoveries compared to those of the corresponding bulk material.
6 For example, Kenneth
et al.7 have shown that reducing the terminal sugar in a dextran coating of iron oxide nanoparticles has had an important impact on the stability of the particles while retaining the same magnetic properties. The magnetic behavior and size dependence of magnetic nanoparticles is controlled by the domain structure of the nanoparticles.
8 The factors that influence the critical size of the single domain consist of the shape of the particles, strength of the crystal anisotropy, value of the magnetic saturation,
and domain wall energy. With increasing values of the radius, magnetic nanoparticles can be classified as superparamagnetic, single domain and multi-domain. The domain wall in magnetic particles is important, because it has a major impact on the magnetization of the particles. In general, the magnetic moments of the particles will scale with the volume of the particle, but because of the presence of domain walls, this scaling is abruptly halted when multiple magnetic domains are formed in a single particle. The critical radius (
rc) represents the point where it is energetically favored for the magnetic particle to exist without a domain wall:
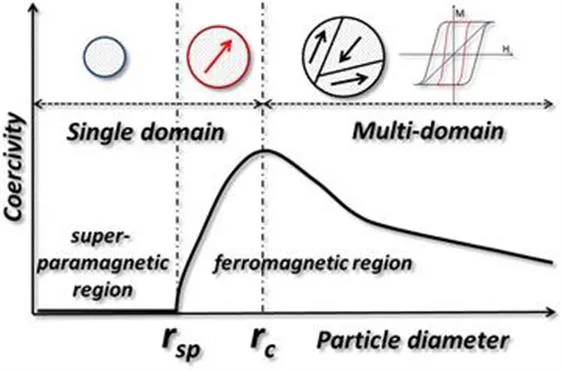
9 where σ represents the magnetic moment per unit mass and Ms2 is the saturation magnetization. In Figure 1.1 it is shown that the coercivity varies dramatically with the particle diameter, unlike the magnetization. In the multi-domain region, it can be observed that the coercivity decreases with the increase of the particle diameter (due to subdivision into domains), while in the single domain region it increases with the radius of the particle. The calculated critical radii for some common magnetic material are: Fe3O4 (4 nm), Co (8 nm), Fe (1 nm) and Ni (35 nm). In other words, a typical colloidal synthetic method is able to produce magnetic nanoparticles that fall into the single domain region. The single domain region can be further divided into two subregions: the superparamagnetic region, where the coercivity is 0 due to the randomizing effects of the thermal energy, and the ferromagnetic region, where the coercivity increases dramatically with the particle diameter. In the absence of a magnetic field, the net magnetic moment is 0 for superparamagnetic nanoparticles, while in an applied magnetic field there will be a net alignment of the magnetic moments. This phenomenon is like paramagnetism with the exception that the magnetic moment originates from a single domain particle (105 atoms) and not from the magnetic moment of a single atom, as observed in paramagnetism. This also means that the saturation magnetization is typically larger in those materials exhibiting paramagnetic behavior.
Figure 1.1 The change in coercivity of magnetic particles as a function of size.
There are multiple ways in which magnetic nanoparticles can be classified based on their properties. In this chapter, we will concentrate on biological applications. First, magnetic nanoparticles must be biocompatible and non...