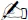![]()
Part I
Experimental Puzzles and Birth of a New Constant in Physics
![]()
1
From Waves to Particles
The content of this chapter will help you understand the shape of the fossil cosmic background radiation,
photoelectricity (in printers and on the Moon), and related topics.
This chapter explains how, on the basis of a set of counter-intuitive experimental results, the scientific community came to invent the notion of photon. Primarily introduced as a mere mathematical trick, the photon’s actual role in many aspects of electromagnetic interaction with matter had to become an ontological fact.
We will thus explain how the full black-body radiation spectrum could only be accounted for by lifting the unnecessary constraint of continuity on possible mean energies carried by an electromagnetic field. This will be shown to be fully compatible with the frequency dependence of electron emission by a metal upon UV light irradiation. Finally, the assimilation of an electromagnetic energy bunch to a particle, travelling in vacuum at light speed, will be firmly established when it appears obvious that it does indeed show every single property to satisfy conservation laws of classical collision theory.
1.1.Short Wavelength Issue in Black-Body Radiation
Observation teaches us that every physical object brought to a temperature T radiates an electromagnetic wave. To express that it has reached a particularly high temperature, we say that it has been heated to “red” or even to “white”, for example. The connection between a temperature and the radiated spectrum colour is thus entrenched in common knowledge. The usual (and classical) model upon which one relies to explain such a phenomenon is as follows: the heat, which is supplied to the system, increases the motion of particles. Locally, accelerated charges act as many antennas that emit an electromagnetic field in an incoherent manner, owing to their random and non-harmonic motion. This willingly partial and very approximate description only serves to justify the fact that the observed spectrum of radiation is continuous, unlike the emission lines from an antenna powered by a periodic electrical current. In parallel to this emission process, the absorption of an electromagnetic wave can be regarded as an excitation of tiny oscillators. The absorption and emission processes then clearly appear to be totally symmetrical.
By convention, the idiom black-body is used to describe a total (perfect) absorber: it absorbs all incident electromagnetic radiation without reflecting any of it. Its total temperature is kept to a fixed value T as a result of a luminance balance. At thermal equilibrium, all absorbed energy is converted into electromagnetic radiation. By definition, an ideal black-body emits radiation, the spectrum of which solely depends on its equilibrium temperature.
The simplest example, which was carefully studied by Wien, is that of a completely sealed oven. One of its walls is punched with a small hole. Radiation power, generated by thermal motion of the atoms and molecules inside, escapes the enclosure through this tiny aperture. The key assumption of such an ideal black-body source is that the chemical nature of the materials coating the cavity’s walls does not play any role in the radiated electromagnetic spectrum.
Following Kirchhoff’s first experiments (1859), Stefan noticed in 1879 that the energy emitted per second, and per unit area, varies in proportion to temperature to the fourth power. He very simply formalizes this empirical law in the form:
where σ is the Stefan–Boltzmann constant. This formula is known as Stefan’s law and today’s σ experimental value is set to 5.67 × 10–8 Wm–2 K–4.
With his punched oven device, supplemented with a spectrometer, Wien (1894) could then observe that the wavelength at which the energy flux is maximal varies as the inverse of the black-body’s equilibrium temperature. It is Wien’s displacement law:
Question 1.1: Black-body orders of magnitude.
1.What is the temperature order of magnitude for a “red hot” material?
2.What is the wavelength at which the spectrum emitted by a black-body at room temperature is expected to be maximum?
Answer:
1.Firstly, it is important to ensure that the object radiates as a black body. Then, according to Wien’s displacement law, if red corresponds to a wavelength of the order of 600 nm, we find: T ≈ 2.9/(0.6 × 10–3) ≈ 5000 K.
Curiously, we know that this is not the colour that one observes at such a temperature. An incandescent lamp filament is about 3000 K. It should be borne in mind that the spectrum is broad. Many other wavelengths are thus also detected and, in particular yellow and blue (even small proportions), come to significantly change the colour perceived by the eye. Therefore, to see the red, it is not the maximum of the spectrum that is to be at 600 nm but a relatively small part of the spectrum. The main part of the spectrum is hence in the infrared range (which we cannot see with the naked eye). A temperature between 600 K and 800 K is sufficient.
2.At 300 K, Wien’s displacement law states λmax ≈ 10 μm. The maximum is then in the near/mid infrared.
These findings push Rayleigh to model the black-body radiation phenomenon on the basis of electromagnetic standing waves physics. His idea, following the concept of the thermodynamics equipartition theorem, is to attribute (kBT)/2 for each degree of freedom of the problem, where kB is the Boltzmann constant.1 For him, it is only a problem of waves. Following Helmholtz and Maxwell, and similar to a guitar string, he imagines that only a (transverse) standing waves system can permanently be set in the above-mentioned cavity. Each of these waves represents a degree of freedom. By counting permitted wave numbers in the cavity (see Question 1.2), it is then possible to obtain the law of Rayleigh–Jeans for spectral energy (per unit volume) radiated from the black-body:
or, with λ = c/v
Question 1.2: Counting electromagnetic modes in a cavity. Calculate the number of electromagnetic modes per unit volume of the cavity in a frequency interval
dv. Assume that the walls are perfectly reflective. Deduce, from the equipartition energy theorem, an electromagnetic energy spectrum expression as a function of frequency and temperature.
Answer: Let us consider one dimension only to start with. Let L be the cavity size. In steady state, the wave must cancel at...