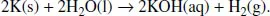![]()
CHAPTER 1
THE PARTICULATE NATURE OF MATTER
1.List the essential differences between a chemical and a physical change. Indicate what type of changes take place in the following process and explain clearly in each case:
Explanation:
During a physical change, the physical property of matter, such as temperature, pressure, density, mass, volume, color, boiling point, melting point, energy content, etc., has changed BUT the chemical property of the matter stays intact. Such physical change is reversible by changing the physical conditions, such as temperature and pressure, back to their original states.
During a chemical change, the chemical property of matter has changed. This change results in the formation of new substances which have different chemical compositions from the starting substance. Such chemical change is usually irreversible by any simple change of the physical conditions.
| Q | What is a chemical property? |
A:The chemical property of a matter is actually its unique chemical potential in reacting with other matter. For example, when sodium reacts with chlorine to form sodium chloride, sodium atom loses an electron while chlorine atom gains an electron. The potential of the sodium atom to lose an electron in the presence of the chlorine atom is the chemical property for the sodium, while the potential to gain an electron for the chlorine atom in the presence of the sodium atom is the chemical property for chlorine.
| Q | So, does that mean that chlorine atom would forever have the same potential to gain electrons irrespective of the type of substances that it reacts with? |
A:No! When a chlorine atom reacts with a hydrogen atom, the chlorine atom does not gain an electron. In fact, the chlorine atom shares electrons with the hydrogen atom. In essence, the chemical property of a matter can vary, depending on the chemical property of the other matter that it is reacting with. You can refer to Understanding Basic Chemistry by K.S. Chan and J. Tan for more details.
Do you know?
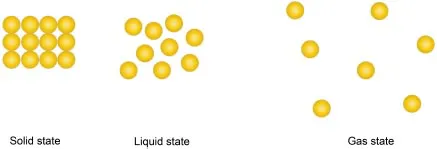
—Matter is made up of very small particles, such as an atom, ion, or molecule, being attracted to one another by electrostatic forces of attraction. The different strengths of the attractive forces between these small particles result in the different physical states of matter.
—There are three states of matter: solid, liquid, and gas. The strength of the attractive force between the particles would give rise to the physical state of the matter.
—If the strength of attraction is very strong, we have solid matter, which results in its fixed shape and fixed volume. A weaker strength of attraction gives us liquid, which has a fixed volume but no fixed shape. The strength of attraction is the weakest in a gas, causing it to have neither a fixed shape nor a fixed volume.
—The force that causes the small particles to be attracted together is known as electrostatic force. It is just a type of “electrical force” between particles that possess opposite “electrical charges.” This electrostatic force is the chemical bond that holds the particles together.
—Thus, if the chemical bond is of different strengths, it will result in different types of arrangement of particles in the physical state, which would therefore decide whether the matter has fixed shape or fixed volume. And because of the different types of arrangement, the particles have different types of motion in the physical state and also different levels of compressibility. Furthermore, the reason for different amounts of energy being involved in the chemical reaction is due to the reaction of different types of matter possessing different chemical bond strengths.
—In the solid state, the particles can only vibrate about a fixed position. But in the liquid and gaseous states, the particles have translational motion; it is free to move randomly in all directions.
| Q | Why is the compressibility of the solid and liquid so much lower than that for the gas? |
A:Since the gas particles are far apart, when we compress a gas by exerting a pressure, the distance of separation between the gas particles can be decreased. If the pressure is high enough, the gas would be converted to the liquid state in which the particles are closer to each other. This is what happens during the liquefaction of gas by the application of pressure. Further compression would then be difficult to make the particles in the liquid state to come even closer together as there would be too much repulsive forces acting between the particles. We could expect more difficulty in compressing a solid than a liquid.
| Q | So, it is actually the weak attractive forces between the particles in the gaseous state that help to “pull” the particles even closer together during compression? |
A:You are right! A lot of students think that it is the applied pressure ALONE that helps to “push” the particles together; this is INCORRECT. Without the attractive forces acting between the particles in the gaseous state, the particles would not be “held on” together in the liquid state. This also explains why a gas with extremely weak attractive forces is difficult to be liquefied through the using of high pressure alone.
(a)Addition of potassium to water.
Explanation:
Potassium reacts with water to give potassium hydroxide and hydrogen as follows:
Since the new products, KOH and H2, have different chemical properties from the reactants, the above change is a chemical change. In addition, since the reactants cannot be formed back from the products simply by the change of the reaction conditions, such as temperature and pressure, the reaction is an irreversible one.
Do you know?
—Potassium is a metal and the fact that hydrogen gas is formed when potassium reacts with water, indicates that water is acting as an acid.
—An acid is characterized by any one of the following three possible reactions:
•An acid reacts with a metal to give hydrogen gas.
•An acid reacts with a carbonate/hydrogencarbonate to give carbon dioxide gas.
•An acid reacts with a base to give salt and water.
(b)Salt dissolves in water.
Explanation:
Salt dissolves in water to give a salt solution. There is no change in the chemical composition of the salt and the water. When heat is applied to the salt solution to drive away the water, the original salt can be recovered. Thus, the dissolving of the salt in water is just a physical change.
| Q | But when the salt is dissolved, it disappears. So, shouldn’t the change be a chemical one? |
A:In a chemical change, the chemical property of the substance will change. Solid salt, such NaCl, consists of sodium (Na+) and chloride (Cl–) ions being attracted to each other. When the salt dissolves, there is no change in the chemical composition except that the ions are simply separated by the water molecules. That is, the Na+ and Cl– ions of the salt are not transformed into any other species that are different from themselves. During evaporation, the water molecules that are between the ions are removed and this process causes the ions to be closer to each other once again. Hence, the dissolution of salt is a physical change!
Do you know?
—The salt that dissolves in water is known as the solute, while the water is known as the solvent.
—The salt solution is homogeneous in nature as you cannot differentiate the salt from the water and the solution does not resemble the solid salt at all. Thus, these may mean that the salt solution is a compound. But we are able dissolve as much salt as possible until it does not dissolve any more. This would mean that the composition of the salt solution is variable. From this, we can conclude...