![]()
Section 1
DIAGNOSTIC PARASITOLOGY
Collection and Submission of Samples
Diagnosis of parasitic infections depends on several factors, such as collection of the sample, transport of the sample to the laboratory, and method of laboratory evaluation. Diagnostic stages of most parasites can be detected in feces, blood, sputum, or skin scrapings. However, infections of immature parasites and latent and occult infections present a diagnostic challenge. Important factors to be considered in the diagnosis of parasitism and the interpretation of results are:
1. Age of the host,
2. Previous exposure to parasites (resistance),
3. Time of the year (spring rise),
4. Physiological relationship (pariparturient rise),
5. Geographical location,
6. Previous use of anthelmintics,
7. History of clinical disease, and
8. Other considerations.
Proper collection and submission of samples to the laboratory increase the accurate diagnosis of parasitic infection.
Fecal Samples
Feces must be fresh for accurate results. As feces age, a diagnosis is complicated because many parasite eggs develop and hatch into larvae. Contaminants such as free-living soil nematodes, fly larvae, mites, and other arthropods often invade feces and complicate a diagnosis.
At least 10 g of fresh feces should be collected. If samples are more than two hours old, samples should be stored at 4°C until examined. Many parasite stages can be stored at 4°C for at least two months with minimal development. For routine shipment to the laboratory, samples can be cooled to 4°C and then packed with ice or other coolant (blue ice) for shipment via any of the 24- to 48-hour transport services. Fecal samples are best stored and sent in whirl-pak bags, small plastic sandwich bags, plastic containers, disposable laboratory gloves turned inside out, or rectal palpation gloves turned inside out. All samples should be clearly labeled with a black indelible marker with the number of the animal, date, and the person responsible for the sample.
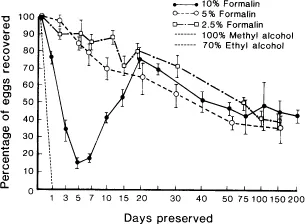
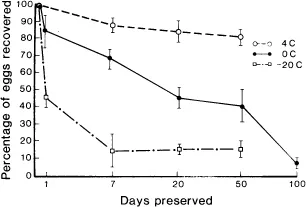
If coolants are not available, samples can be stored indefinitely in 10% formalin (one part feces, nine parts 10% formalin), but limitations must be noted (Fig. 1). Approximately 50% of the ruminant strongyle eggs were detected in feces stored in 10% formalin for 200 days. Storage by freezing is very inefficient, and storage in 70% ethyl alcohol or 100% methyl alcohol is unacceptable (Fig. 2).
When Giardia sp. is suspected, feces can be placed in polyvinyl alcohol fixative at a ratio of one part feces to two parts polyvinyl alcohol or in 5% formalin for fixation and transport. Also, fecal smears on slides can be stained with Gormori’s trichrome, iron-hematoxylin, clorzol black, or Giemsa stains and submitted to the laboratory in standard slide mailers.
Fig. 1. Effect of formalin and alcohol preservation on mean recovery of strongyle eggs over time. Vertical lines indicate 1 SD. From Foreyt W.J.: Recovery of nematode eggs and larvae in deer: Evaluation of fecal preservation methods. J Am Vet Med Assn 189:1065-1067, 1986. Reprinted with permission.
Fig. 2. Effect of temperature on the mean recovery of strongyle eggs from deer feces over time. From Foreyt W.J.: Recovery of nematode eggs and larvae in deer: Evaluation of fecal preservation methods. J Am Vet Med Assn 189:1065-1067, 1986. Reprinted with permission.
Fecal Flotation
For normal stools, the usual diagnostic test is the fecal flotation test. This test concentrates eggs and oocysts present in the feces into a drop of solution for easy identification and enumeration of parasitic stages. Important comparative factors in the fecal flotation technique are the specific gravity of the flotation solution, the viscosity or type of solution used, and the rate of plasmolysis caused by the solution. A specific gravity that is too low will not float many stages, whereas a solution with a specific gravity that is too high will cause plasmolysis, osmosis, or rupture of the stages, making diagnosis difficult (Fig. 3). Also, as the specific gravity is increased, excessive debris also floats, which decreases the efficiency of the test. Most parasitic stages float efficiently at a specific gravity of 1.2 to 1.3. My laboratory uses sugar solution (specific gravity = 1.27). Sugar has a distinct advantage over salt solution in that less plasmolysis and distortion occur in the eggs and oocysts. Fecal-flotation slide preparations from sugar solution can be kept at 4°C for at least 24 hours, and often for several weeks to months, with a minimum of distortion of eggs. These slides can be used as next-day reference and teaching slides. Disadvantages of sugar solutions are that sugar can be messy and sticky, and sugar attracts flies and other arthropods. When salt solutions are used, egg distortion usually occurs in a few hours, and the slides have a tendency to crystallize and dry out very quickly. To increase the shelf life of a fecal-flotation slide, one can put fingernail polish or quick-drying glue around the coverslip. Freezing the prepared slide is often effective for preserving the material for many months or years. One additional advantage of sugar solution is that some parasite eggs, such as the salmon poisoning fluke, Nanophyetus salmincola, float better in sugar than salt solutions. In salt solutions, the eggs often do not float and are often missed during examination.
Blood
Blood is used for the diagnosis of specific pa...