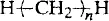![]()
CHAPTER 1
INTRODUCTION TO POLYMER SCIENCE
Polymer science was born in the great industrial laboratories of the world of the need to make and understand new kinds of plastics, rubber, adhesives, fibers, and coatings. Only much later did polymer science come to academic life. Perhaps because of its origins, polymer science tends to be more interdisciplinary than most sciences, combining chemistry, chemical engineering, materials, and other fields as well.
Chemically, polymers are long-chain molecules of very high molecular weight, often measured in the hundreds of thousands. For this reason, the term “macromolecules” is frequently used when referring to polymeric materials. The trade literature sometimes refers to polymers as resins, an old term that goes back before the chemical structure of the long chains was understood.
The first polymers used were natural products, especially cotton, starch, proteins, and wool. Beginning early in the twentieth century, synthetic polymers were made. The first polymers of importance, Bakelite and nylon, showed the tremendous possibilities of the new materials. However, the scientists of that day realized that they did not understand many of the relationships between the chemical structures and the physical properties that resulted. The research that ensued forms the basis for physical polymer science.
This book develops the subject of physical polymer science, describing the interrelationships among polymer structure, morphology, and physical and mechanical behavior. Key aspects include molecular weight and molecular weight distribution, and the organization of the atoms down the polymer chain. Many polymers crystallize, and the size, shape, and organization of the crystallites depend on how the polymer was crystallized. Such effects as annealing are very important, as they have a profound influence on the final state of molecular organization.
Other polymers are amorphous, often because their chains are too irregular to permit regular packing. The onset of chain molecular motion heralds the glass transition and softening of the polymer from the glassy (plastic) state to the rubbery state. Mechanical behavior includes such basic aspects as modulus, stress relaxation, and elongation to break. Each of these is relatable to the polymer’s basic molecular structure and history.
This chapter provides the student with a brief introduction to the broader field of polymer science. Although physical polymer science does not include polymer synthesis, some knowledge of how polymers are made is helpful in understanding configurational aspects, such as tacticity, which are concerned with how the atoms are organized along the chain. Similarly polymer molecular weights and distributions are controlled by the synthetic detail. This chapter starts at the beginning of polymer science, and it assumes no prior knowledge of the field.
1.1 FROM LITTLE MOLECULES TO BIG MOLECULES
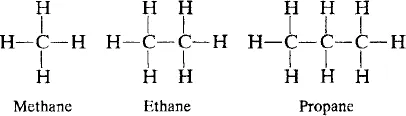
The behavior of polymers represents a continuation of the behavior of smaller molecules at the limit of very high molecular weight. As a simple example, consider the normal alkane hydrocarbon series
These compounds have the general structure
where the number of —CH2— groups, n, is allowed to increase up to several thousand. The progression of their state and properties is shown in Table 1.1.
Table 1.1 Properties of the alkane/polyethylene series
| 1–4 | Simple gas | Bottled gas for cooking |
| 5–11 | Simple liquid | Gasoline |
| 9–16 | Medium-viscosity liquid | Kerosene |
| 16–25 | High-viscosity liquid | Oil and grease |
| 25–50 | Crystalline solid | Paraffin wax candles |
| 50–1000 | Semicrystalline solid | Milk carton adhesives and coatings |
| 1000–5000 | Tough plastic solid | Polyethylene bottles and containers |
| 3−6 × 105 | Fibers | Surgical gloves, bullet-proof vests |
At room temperature, the first four members of the series are gases. n-Pentane boils at 36.1°C and is a low-viscosity liquid. As the molecular weight of the series increases, the viscosity of the members increases. Although commercial gasolines contain many branched-chain materials and aromatics as well as straight-chain alkanes, the viscosity of gasoline is markedly lower than that of kerosene, motor oil, and grease because of its lower average chain length.
These latter materials are usually mixtures of several molecular species, although they are easily separable and identifiable. This point is important because most polymers are also “mixtures”; that is, they have a molecular weight distribution. In high polymers, however, it becomes difficult to separate each of the molecular species, and people talk about molecular weight averages.
Compositions of normal alkanes averaging more than about 20 to 25 carbon atoms are crystalline at room temperature. These are simple solids known as wax. It ...