The author addresses the principles of the subject from the viewpoint of modern processes, emphasising a general science narrative approach in the main text, with quantitative background derived in enabling 'cookie' appendices. The book ends with an innovative chapter dealing with how sedimentology is currently informing a variety of cognate disciplines, from the timing and extent tectonic uplift to variations in palaeoclimate. Each chapter concludes with a detailed guide to key further reading leading to a large bibliography of over 2500 entries. The book is designed to reach an audience of senior undergraduate and graduate students and interested academic and industry professionals.

- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
The sedimentary record on Earth stretches back more than 4.3 billion years and is present in more abbreviated forms on companion planets of the Solar System, like Mars and Venus, and doubtless elsewhere. Reading such planetary archives correctly requires intimate knowledge of modern sedimentary processes acting within the framework provided by tectonics, climate and sea or lake level variations. The subject of sedimentology thus encompasses the origins, transport and deposition of mineral sediment on planetary surfaces.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Part 1
MAKING SEDIMENT
… the soil which has kept breaking away from the high lands during these ages and these disasters, forms no pile of sediment worth mentioning, as in other regions, but keeps sliding away ceaselessly and disappearing in the deep.
Plato, Critias, Vol. 9, Loeb Classical Library
Introduction
The noun sediment comes to the English language from the Latin root sedimentum, meaning settling or sinking down,aform ofthe verb sedere,to sit orsettle. In earth and environmental sciences, sediment has a wide context that includes many forms of organic and mineral matter. In Part 1 we look more deeply at the origins of the sediment that occurs on and under the surface of the solid planets and which may be used to infer past environmental conditions and changes. Sediment accumulations may be grandly viewed as the great stratal archive of past surface environments, or more basically as‘dirt’. There has been sediment on the surface of the Earth since the Archaean, with the oldest known sediment grains dating from at least 4.4Ga (Part 1 Fig. 1). Sediment also mantles the surface of many other planets and their satellites, notably Mars, Venus and Saturn’s moon, Titan.
In scientific usage, Earth’s sediment is best divided into three end-members:
- clastic—originating from pre-existing rock outside a depositional area as transported grains, the commonest being mineral silicate grains, known widely as siliciclastic sediment;
- chemical—being the result of inorganic or organically mediated chemical precipitation within the depositional area;
- biological—derived from skeletal material associated with living tissues.
These simple divisions are robust enough to include even the highly esoteric sediment forms that are turning up in the wider Solar System, like the solid ice particles transported and deposited by liquid methane on Titan.Ofcourse there are unusual, hybridormixed origins for some sediment but these can easily be accommodated (e.g. bioclastic, volcaniclastic). Note that the classification is restricted to grains that were sedimented; there are sedimentary horizons in the stratigraphic record that originated as precipitates below the deposited sediment surface, often bacteri-ally controlled. These were never sedimented as such and are considered as secondary or diagenetic sediments that post-dated physical deposition of host primary sediment. Deposited sediment accumulates as successive layers, termed strata, and such deposits as a whole are said to be stratified. The succession of strata in any given deposit is controlled by environmental factors and their correct interpretation involves a deep understanding of how present and past environments have evolved over time.
The chemical and biochemical processes that produce sediment also give other soluble byproducts; these chemical species control oceanic and atmospheric composition and provide long-term sourcing for base cations that nourish plant life and counteract acid deposition in temperate forested catchments. Chemical earth-surface processes have undoubtedly changed over deep geological time, in response to atmospheric and hydrological changes, whilst biological processes have changed hand-in-hand with organic evolution. By this view, sediment production is an accident of weathering and evolution—a waste product. Ever since the Archaean, the planet has ‘learnt’ how to cope with this waste, just like it has with the waste oxygen produced during plant photosynthesis. There was no predeterminism associated with the processes of sediment production on early Earth or any other planet. Sediment simply fell out (forgive the pun) of the rock cycle in which primary rock is chemically and physically altered. Compare for example sediment on the Moon with the Earth.In the former the sediment is a fragmented remnant from past meteoritic impact events. In the latter sediment is highly varied in its origins, composition, size and physical properties. Its role as an accidental part of the Rock Cycle establishes sedimentology as a fundamental part of Earth System Science. Indeed, as one nice semi-popular review entitled it a decade ago (Stanley & Hardie, 1999), from the point-of-view of calcareous sediment, ‘Hypercalcification: Paleontology Links Plate Tectonics and Geochemistry to Sedimentology’. We shall examine such grand claims later in this book (Chapter 23).
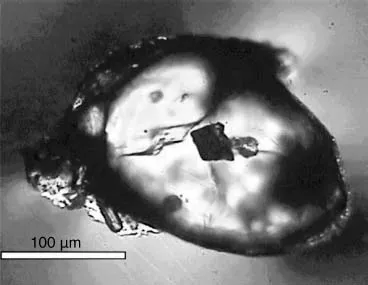
Part1 Fig. 1 Image of the famous Jack Hills’zircons from the Cryptic Era of the Hadean Eon (discovery paper of Wilde et al., 2001). The location is ∼800 km north of Perth in Western Australia in the Narryer Gneiss Terrane of the Yilgarn Craton, a group of folded and metamorphosed supracrustal rocks thought to have originally contained sedimentary siliciclastic rocks. Detrital zircons with ages greater than 4 billion years old occur here, the oldest being 4.4 Ga (±8 million), the oldest dated material originating on Earth. The source is a metamorphosed conglomerate considered to have an age ∼3.0Ga, so the detrital zircons are sourced from pre-existing rocks, probably subduction zone plutonic igneous intrusions which were then weathered and the resultant sediment deposited as sedimentary rock. The zircons are evidence for the existence of continental-type crust on the surface of the Earth during the Hadean Eon, contrasting with earlier ideas on the earliest phase of Earth’s history in which continental crust was thought absent and plate tectonics inoperative until much later. Additionally, oxygen isotopic ratios in the zircons provide evidence for the presence of liquid water on the Earth’s surface at this time. The image isa general photograph of a pristine Jack Hills’zircon (Curtin University website). Longest axis of crystal ∼250μm.
Chapter 1
CLASTIC SEDIMENT AS A CHEMICAL AND PHYSICAL BREAKDOWN PRODUCT
Few ken to whom this muckle monument stands, Some general or admiral I’ve nae doot, On the hill-top whaur weather lang syne Has blotted its inscribed palaver oot.
Hugh MacDiarmid, ‘The Monument’, 1936, Complete Poems, Vol. 1, Carcanet, 1993.
1.1 Introduction: clastic sediments—‘accidents’ of weathering
Terrestrial clastic sedimentary rocks are usually quite different in their composition from the igneous and metamorphic rocks that sourced them. This is because they are derived from an altered regolith with a soil profile produced by chemical weathering of pristine bedrock and the source of mineral grains for such sediment. For example, feldspar is the commonest mineral in bedrock of the Earth’s continental crust (about 60% of the total) but quartz is usually predominant in clastic sediments and sedimentary rocks. Despite this difference the principle of conservation of mass tells us that for all elements present in the exposed crust and released by weathering, exactly the same levels of abundance must occur in the average total sedimentary mass. Thus the average chemical composition of all sediments is roughly that of the igneous rock, granodiorite, representing the mean composition of middle to upper continental crust.
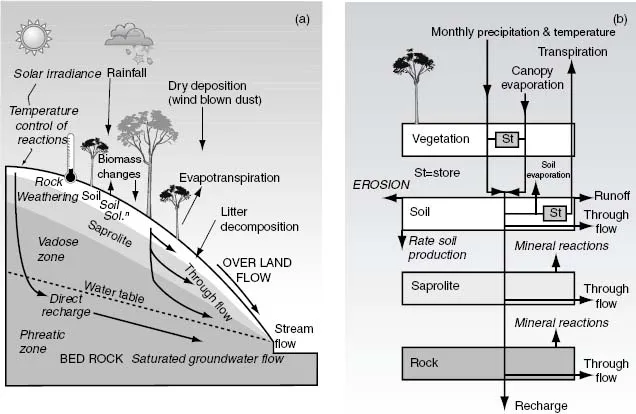
It is traditional to divide rock weathering into physical and chemical components, but in reality the two are inextricably interlinked. Water is the chief reactant and plays a dual role since it also transports away both dissolved and solid weathering products. Earthispresentlyuniqueinits abundanceofwater and water vapour, yet Mars also had an earlier watery prehistory. It is easy to take water for granted, the deceptively simple molecule H2O has remarkable properties of great importance for rock and mineral weathering (Cookie 1). These include its solvent and hydration properties, wetting effects due to high saturation and anomalous decreaseofdensityatlow liquid temperatures and after freezing. An outline of the near-surface terrestrial hydrological cycle is given in Fig. 1.1.
Want to know more about the structure and properties of water? Turn to Cookie 1.
Fig. 1.1 (a) The main components ofthe terrestrial hillslope hydrological cycle. (b) Box model for water budgetinthe surface and near-surface weathering zones.
Chemical weathering involves aqueous reactions with a strong biochemical component since dissolved atmospheric gases are aided by soil-generated gases, dilute acids and organic ligands. Further, the reactions are complex since silicate minerals are involved, with their many constituent anions and cations; also the amount of water and dissolved ions varies in both time and space. Four main mechanisms contribute to chemical weathering: dissolution, oxidation, hydrolysis and acid hydrolysis. Reactions usually occur at mineral surfaces in the unsaturated (vadose) zone where, close to the local Earth’s surface, rock pores contain atmospheric gases, water, living and dead vegetation and bacteria—all play an important role in weathering. The result is a regolith and soil profile whose characteristics depend upon climate and rock type.
Physical weathering involves the application of differential stresses to rock and mineral discontinuities in the unsaturated zone. These cause fragmentation and are due to erosional unloading, gravity, wind shear, salt crystallization from groundwaters, freeze–thaw and differential thermal expansion.
The combined effects of biochemical and physical weathering produce a weathered regolith profile in bedrock that comprises:
- surface soil with significant living and dead organic content;
- chemically altered rock termed saprolite that nevertheless preserves its framework coherency without volume loss (isovolumetric weathering);
- chemically unaltered but often physically fragmented (exfoliated) bedrock.
Interfaces between these layers are in a state of slow downward motion as the landscape reduces. In fact, landscape dating by cosmogenic isotopes and other means reveals that a steady-state system often exists, with the material mass removed by erosion being replaced by an equal volume made available from below for further decomposition.
Weathering acts on:
- mineral aggregates at or close to Earth’s surface with their many intracrystalline pores and fracture networks;
- imperfect crystals with surface and lattice defects.
Weathering involves:
- bond breaking, physically by cracking and chemically by solution (see Cookie 2);
- broken bond (danglers) adoption by ionic predators;
- electron removal from the easily stripped transition metals like Fe and Mn (see Cookie 3).
Weathering depends on:
- H2O throughput or hydraulic conductivity by laminar water flow in pore spaces between minerals, microporosity within minerals and cracks across minerals;
- H2O dissociation into the reactive H and OH ions (see Cookie 1);
- CO2 concentration via atmospheric and soil processes;
- temperature, which controls both reaction rates (Fig. 1.2) in silicate minerals via the results of the Arrhenius principle (see Cookie 3 and further below) and the rate of aqueous throughput via its control on water’s dynamic viscosity.
Fig. 1.2 Arrhenius plot to show that the flux of Si from small, simple-geology granite catchments depends on temperature,once corrections are madetoallow for rainfall amount. This is because the rate constant is determined by the Arrhenius effect discussedinthe main text and Cooki...
Table of contents
- COVER
- TITLE
- COPYRIGHT
- PREFACE
- ACKNOWLEDGEMENTS
- Part 1: MAKING SEDIMENT
- Part 2: MOVING FLUID
- Part 3: TRANSPORTING SEDIMENT
- Part 4: MAJOR EXTERNAL CONTROLS ON SEDIMENTATION AND SEDIMENTARY ENVIRONMENTS
- Part 5: CONTINENTAL SEDIMENTARY ENVIRONMENTS
- Part 6: MARINE SEDIMENTARY ENVIRONMENTS
- Part 7: ARCHITECTURE OF SEDIMENTARY BASINS
- Part 8: TOPICS: SEDIMENT SOLUTIONS TO INTERDISCIPLINARY PROBLEMS
- COOKIES
- MATHS APPENDIX
- REFERENCES
- COLOUR PLATES FALL BETWEEN
- INDEX
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Sedimentology and Sedimentary Basins by Mike R. Leeder in PDF and/or ePUB format, as well as other popular books in Physical Sciences & Geology & Earth Sciences. We have over 1.5 million books available in our catalogue for you to explore.