This comprehensive update of a classic text presents the latest research on the dental pulp and its interaction with other tissues, highlighting its central role in both local and systemic health. The second edition has been completely revised to incorporate new chapters featuring the most topical issues in research and clinical practice, including developments in stem cell research and pulpodentin regeneration, the effects of the aging process on the pulp, and the interdependent relationship of the pulp and restorative dental procedures. New contributors bring fresh perspective to topics such as pulpal infections, odontalgia, and the relationship between the pulp and periodontal disease. Each chapter provides an introduction to its major themes for the busy clinician or dental student as well as up-to-date, biologically based clinical recommendations for restorative and endodontic procedures. Practicing clinicians will find this information to be essential to providing accurate diagnoses and effective treatment.

eBook - ePub
Seltzer and Bender's Dental Pulp
Second Edition
- 512 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information

Development of the Pulpodentin Complex
Rena D’Souza, DDS, MS, PhD
Chunlin Qin, DDS, MS, PhD
Chunlin Qin, DDS, MS, PhD
Dentin is a unique, avascular mineralized connective tissue that forms the bulk of the tooth. It underlies enamel in the crown and cementum in the roots, providing these tissues structural support and the tooth its resilience. In a mature tooth, dentin encloses a richly innervated and highly vascularized soft connective tissue, the dental pulp. Dentin and pulp are derived from the dental papilla, whose cells migrate to the first branchial arch from within the ectomesenchyme of the cranial neural crest. The tissues remain closely associated during development and throughout the life of an adult tooth and are hence most commonly referred to as the pulpodentin complex. It is this biologic intimacy that dictates the response of the pulpodentin complex to physiologic and pathologic stimuli.
Because the practice of endodontics involves manipulation of both the dentin and pulp, learning about the mechanisms that lead to their formation is crucial and will create a better understanding of the response to and treatment of pulpal injuries. The purpose of this chapter is to provide a background for the succeeding chapters that discuss the biology of the mature pulpodentin complex during health and disease and in aging. The chapter has two goals: The first goal is to review classic and current knowledge of the events in tooth development that lead to odontoblast differentiation and to convey the excitement of this flourishing field. Attention is focused on the common themes that have emerged and what is known about the influence of tooth-signaling molecules and transcription factors on the development and homeostasis of the pulpodentin complex. The second goal of this chapter is to describe the general principles of dentin matrix formation, particularly the synthesis and secretion of extracellular matrix molecules and their postulated roles in the biomineralization of dentin. Examples of how basic information about the normal biology of the pulpodentin complex can be applied toward solving clinical problems are integrated throughout the chapter. The overarching goal is to emphasize how fundamental theories about development and homeostasis of differentiated and undifferentiated or stem cell populations can be translated to regenerative approaches targeted at restoring the integrity of the adult pulpodentin complex.
Tooth Development (Odontogenesis)
For several decades, the developing tooth organ has served as a valuable paradigm to study the fundamental processes involved in organogenesis. These processes are (1) the determination of position, through which the precise site of tooth initiation is established; (2) the determination of form or morphogenesis, through which the size and shape of the tooth organ are set; and (3) cell differentiation, through which organ-specific tissues are formed by defined cell populations, each with unique properties. The dental literature is enriched with excellent reviews on tooth development, and the reader is encouraged to study the topic in further detail.1–4
General features
Although the tooth is a unique organ, the principles that guide its development are shared in common with other organs such as the lung, kidney, heart, mammary glands, and hair follicles.5,6 The most important among developmental events are those guiding epithelial-mesenchymal interactions, which involve a molecular crosstalk between the ectoderm and mesenchyme, two tissues that have different origins. Although only vertebrates have teeth, their development involves genetic pathways that are also active in invertebrates. This conservation of a “molecular toolbox” for organogenesis throughout evolution proves that certain master regulatory molecules are critical to all tissue interactions during development. New studies have also shown that tooth-signaling molecules are repeatedly used at various stages of development. Both tooth morphogenesis and cell differentiation occur as a result of sequential interactions. Hence, it is not one biologic event involving a single molecule but a series of interactions involving several molecules that leads to the development of the pulpodentin complex.3,4
Importantly, signaling is reciprocal, whereby an exchange of information occurs in both directions, from dental epithelium to mesenchyme and from dental mesenchyme to epithelium. For example, in experiments where dental epithelium was separated from mesenchyme, cusp patterning failed to occur. Similarly, in the absence of dental epithelium, odontoblasts are unable to differentiate from dental mesenchyme.7–9
It is only logical to apply these basic developmental principles to the current understanding of how the adult or mature pulpodentin complex responds to injury and repair. The latter clearly involves a series of molecules that operate in concert to dictate the outcome of pulpal disease and therapies. In the adult situation, whether certain cells and molecules can mimic the inductive influence of dental epithelium during development has yet to be definitively proven and remains a subject of interest in pulpal biology research.
Stages of tooth development
Teeth develop in distinct stages that are easily recognizable at the microscopic level. These stages in odontogenesis, described by the histologic appearance of the tooth organ, are termed, from early to late, the lamina, bud, cap, and bell (early and late) stages of tooth development.10–12 Although the following descriptions use these common terms, the modern literature uses functional terminology to describe odontogenesis in four phases: (1) initiation, (2) morphogenesis, (3) cell differentiation or cytodifferentiation, and (4) matrix apposition (Fig 1-1). The photomicrographs in Fig 1-2 depict the morphologic stages of tooth development.
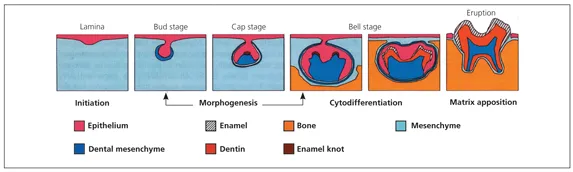
Fig 1-1 Stages of tooth development. Note the sequential transformation from the dental lamina to a distinctly shaped dental organ. The transient appearance of the enamel knot in the region of the forming cusp tips precedes the terminal differentiation of cells and the formation of specialized matrices. (Reprinted from Thesleff and Sharpe12 with permission.)
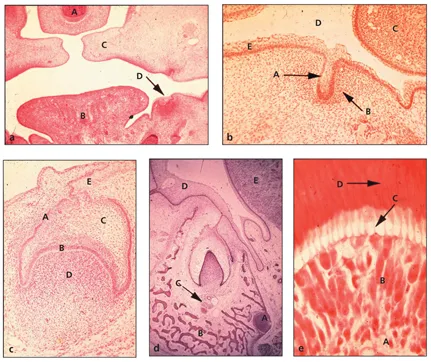
Fig 1-2 Histologic survey of odontogenesis in a pig embryo. (Courtesy of the University of Texas Health Science Center at Houston, Dental Branch.) (a) Lamina stage: A, nasal septum; B, tongue; C, palatal shelves; D, dental lamina (hematoxylin-eosin [H&E] stain; original magnification ×4). (b) Bud stage: A, ectodermal outgrowth; B, dental mesenchyme; C, tongue; D, oral cavity space; E, oral ectoderm (H&E stain; original magnification ×10). (c) Cap stage or transition to early bell stage: A, outer dental epithelium; B, internal dental epithelium; C, stellate reticulum; D, dental papilla ectomesenchyme; E, dental lamina (H&E stain; original magnification ×10). (d) Late bell stage: A, nerve bundle; B, alveolar bone; C, vas-culature; D, oral ectoderm; E, tongue. Note the extension of the dental lamina on the right aspect of the dental organ that will form the succeda-neous incisor (H&E stain; original magnification ×10). (e) Onset of dentinogenesis: A, dental pulp; B, cluster of odontoblasts that appear crowded at the tip; C, odontoblast process; D, dentin (H&E stain; original magnification ×20).
Lamina stage
The dental lamina is the first morphologic sign of tooth development and is visible around 5 weeks of human development and at embryonic day 11 (E11) in mouse gestation. This thickening of the oral epithelium lining the frontonasal, maxillary, and mandibular arches occurs only at sites where tooth organs will develop. At the lamina stage, cells in the dental epithelium and underlying ectomesenchyme are dividing at different rates, more rapidly in the latter. As explained later, ...
Table of contents
- Cover Page
- Cover Page
- Cover Page
- Title Page
- Copyright Page
- Table of Contents
- Preface
- Contributors
- 1. Development of the Pulpodentin Complex
- 2. Formation and Repair of Dentin in the Adult
- 3. Pulpodentin Complex
- 4. Pulp as a Connective Tissue
- 5. Stem Cells and Regeneration of the Pulpodentin Complex
- 6. Circulation of the Pulp
- 7. Dental Innervation and Its Responses to Tooth Injury
- 8. Pain Pathways and Mechanisms of the Pulpodentin Complex
- 9. Pharmacologic Control of Dental Pain
- 10. Pulpal Infections, Including Caries
- 11. Molecular Mediators of Pulpal Inflammation
- 12. Interrelationship of the Pulp and Apical Periodontitis
- 13. Repair of Pulpal Injury with Dental Materials
- 14. Caries, Restorative Dentistry, and the Pulp
- 15. Effects of Thermal and Mechanical Challenges
- 16. Interrelationship of Pulpal and Periodontal Diseases
- 17. Root Resorption
- 18. Aging and the Pulp
- 19. Differential Diagnosis of Toothache: Odontogenic Versus Nonodontogenic Pain
- 20. Interrelationship of Pulp and Systemic Disease
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Seltzer and Bender's Dental Pulp by Kenneth M Hargreaves,Harold E Goodis,Franklin Tay in PDF and/or ePUB format, as well as other popular books in Medicine & Dentistry. We have over 1.5 million books available in our catalogue for you to explore.