1.1 Basics of Light
Light is ubiquitous in our daily life. Without light, there would be no life on the earth. For thousands of years, humans have been fascinated by light phenomena and tried to understand and harness light for better serving their needs. The study of light and its interactions with matter is known as optics, which is an important research field in modern physics, engineering, and life science. Over the past 100 years, many important discoveries about light phenomena and developments in optical theories and technologies have taken place. With the advent and rapid advances of the computer, Internet, and wireless technologies over the past 30 years, we have witnessed the emergence of many new optical technologies and their expanding applications in detection or diagnosis, manufacturing, product processing, communications, energy generation, etc.
This chapter presents an introduction to the basic characteristics of light and optical theories, followed by a brief overview of light scattering technology and its applications to food and agricultural products. In our daily life, the term light often refers to visible light, which only covers a very narrow section of the electromagnetic spectrum. In this book, most of our discussion about light scattering technology is focused on the visible and near-infrared region because light in this spectral region can undergo multiple scattering and long-distance propagation in food and biological materials.
In classical electromagnetic theory, light is treated as electromagnetic radiation, consisting of both electric and magnetic vector waves. The wave characteristics are described by frequency (υ), wavelength (λ), and velocity (c). Light travels in vacuum at a speed of 300,000 km/s, regardless of its frequency or wavelength. However, in a medium, light travels slower than in vacuum; its actual speed depends on the type of waves or wavelengths and the property of the medium. As the speed of light changes from one medium to another, the light no longer travels in a straight direction. Instead, the light changes its traveling direction after it enters the second medium. This important phenomenon is called refraction, which contributes to light scattering and propagation in food and biological materials and will be further discussed in a later section and also in Chapter 2.
Classical electromagnetic theory adequately describes the wave characteristics and most phenomena of light and its interactions with matter, except for a few special situations. Modern quantum theory, on the other hand, considers light to be composed of photons of different frequencies, or small packets of energy. A photon carries electromagnetic energy, but has zero mass at rest. The energy of a photon, E, is proportional to frequency υ (or inversely proportional to wavelength λ), which can be expressed by the following equation:
where E has the unit of joule or J, h = 6.63 × 10−34 J/s is Planck’s constant, υ = c/λ is the frequency in 1/s, and c is the velocity of light in the medium. This wave–photon duality property provides the most complete explanation for virtually all light phenomena that are now known to us, and are widely used in studying the interaction of light with matter, including biological and food materials.
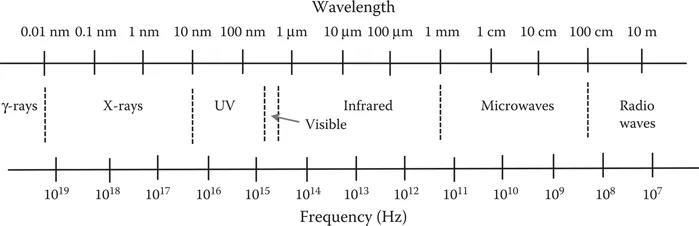
Since the energy of photons is determined by wavelength, photons of different wavelengths carry different levels of energy and thus behave differently when they interact with matter. The electromagnetic radiation emitting from the sun or a black body consists of a spectrum covering a broad range of frequencies (or wavelengths). The electromagnetic spectrum is normally divided into different regions according to wavelengths or frequencies, in the decreasing order of energy: gamma rays, x-rays, ultraviolet (UV) light, visible light, infrared light, microwaves, and radio waves (Figure 1.1). The major characteristics of these radiation regions and their typical applications are briefly discussed in the following subsections.
1.1.1 Gamma Rays and X-Rays
Gamma rays and x-rays are the most powerful electromagnetic radiation in the electromagnetic spectrum. Gamma rays cover a portion of the electromagnetic spectrum with wavelengths of less than 10 pm (1 pm = 10−12 m) or with corresponding frequencies greater than 3 × 1019 Hz, whereas x-rays cover wavelengths between 0.01 and 10 nm (1 nm = 10−9 m) or the frequency range of 3 × 1019–3 × 1016 Hz. Both gamma rays and x-rays are ionizing radiation, which can liberate electrons from atoms or molecules, and can permanently damage or destroy living cells. They can travel in straight lines and their paths cannot be altered by electrical or magnetic fields. Gamma rays and x-rays can usually penetrate deep into matter, although the degree of penetration depends on their energy and the medium they are traveling through. Because of their low absorption and the ability of “seeing through” objects, x-ray-based technologies (i.e., x-ray imaging, computed tomography or CT, etc.) are widely used in medical diagnosis and also in food inspection. Gamma rays are useful in astronomy and physics for studying high-energy objects or regions, the irradiation of food, and diagnostic imaging in medicine.
FIGURE 1.1 Spectrum of electromagnetic radiation from gamma rays to radio waves.
1.1.2 UV Light
UV light refers to the section of an electromagnetic spectrum with the wavelengths approximately between 10 and 400 nm (or ~3 × 1016–8 × 1014 Hz), which is shorter than visible light but longer than x-rays. Sunlight contains UV light, but most of it is absorbed by the ozone layer and atmosphere. UV light can also be produced by electric arcs and light sources such as mercury-vapor lamps, tanning lamps, and black lights. Although not as powerful as x-rays, UV light can heat matter, cause chemical reactions, and make substances to glow or fluoresce, and it can damage living tissues. UV light can be further divided into UV-C (10–280 nm), UV-B (280– 315 nm), and UV-A (315–400 nm). UV-C rays are the most harmful and are almost completely absorbed by the atmosphere. UV-B rays are harmful to living organisms and can damage deoxyribonucleic acid (DNA). UV-A rays can age skin cells and damage DNA, but they also have beneficial effects to living organisms by inducing the production of vitamin D in the skin.
UV light has found many applications, be...