
- 656 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Spinal Cord Injury
About this book
Handbook of Clinical Neurology: Spinal Cord Injury summarizes advances in the clinical diagnosis, monitoring, prognostication, treatment, and management of spinal cord injuries. More specifically, it looks at new and important developments in areas such as high-resolution noninvasive neuroimaging, surgery, and electrical stimulation of motor, respiratory, bladder, bowel, and sexual functions. It also reviews the latest insights into spontaneous regeneration and recovery of function following rehabilitation, with emphasis on novel therapeutic strategies, such as gene therapy, transcranial stimulation, brain-machine interfaces, pharmacological approaches, molecular target discovery, and the use of olfactory ensheathing cells, stem cells, and precursor cells.
Organized in five sections, the book begins with an overview of the development, maturation, biomechanics, and anatomy of the spinal cord before proceeding with a discussion of clinical diagnosis and prognosis as well as natural recovery, ambulation, and function following spinal cord injury. It then examines clinical neurophysiology in the prognosis and monitoring of traumatic spinal cord injury; medical, surgical and rehabilitative management of spinal cord trauma; and some new approaches for improving recovery in patients, including restoration of function by electrical stimulation, locomotor training, and the use of robotics. Other chapters cover cell transplantation, artificial scaffolds, experimental pharmacological interventions, and molecular and combinatorial strategies for repairing the injured spinal cord. This volume should be of interest to neuroscience and clinical neurology research specialists and practicing neurologists.
- Comprehensive coverage of the latest scientific understanding of spinal cord injuries
- Detailed coverage of current treatment best practices and potential future treatments
- Connects leading edge research programs to future treatment opportunities
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Chapter 1
Development and maturation of the spinal cord
Abstract
The human central nervous system (CNS) may be the most complex structure in the universe. Its development and appropriate specification into phenotypically and spatially distinct neural subpopulations involves a precisely orchestrated response, with thousands of transcriptional regulators combining with epigenetic controls and specific temporal cues in perfect synchrony. Understandably, our insight into the sophisticated molecular mechanisms which underlie spinal cord development are as yet limited. Even less is known about abnormalities of this process – putative genetic and molecular causes of well-described defects have only begun to emerge in recent years. Nonetheless, modern scientific techniques are beginning to demonstrate common patterns and principles amid the tremendous complexity of spinal cord development and maldevelopment. These advances are important, given that developmental anomalies of the spinal cord are an important cause of mortality and morbidity (Sadler, 2000); it is hoped that research advances will lead to better methods to detect, treat, and prevent these lesions.
Gross embryology
Overview
A human term pregnancy lasts approximately 40 weeks, and the most dramatic and complex developmental processes are completed in the embryonic period spanning the first 8 weeks. In the embryonic period, critical developmental milestones include establishment of the midline and anteroposterior axis, formation of the three germ layers through gastrulation, and organogenesis. The subsequent fetal period is comparatively simple, wherein the developing human predominantly grows in size.
Fertilization to gastrulation
Fertilization characteristically takes place in the ampullary region of the fallopian tube. The fertilized egg then undergoes a number of mitotic divisions, eventually forming a 16-cell morula 3 days after fertilization (Fig. 1.1). Around the time the morula enters the uterus, it becomes known as a blastocyst and develops a cystic cavity known as a blastocele. By this time, the inner and outer cell masses have formed, which give rise to the embryo proper and the placenta respectively. The outer cell mass, also known as the trophoblast, secretes proteolytic enzymes which facilitate implantation in the endometrium, which occurs about 1 week following fertilization.
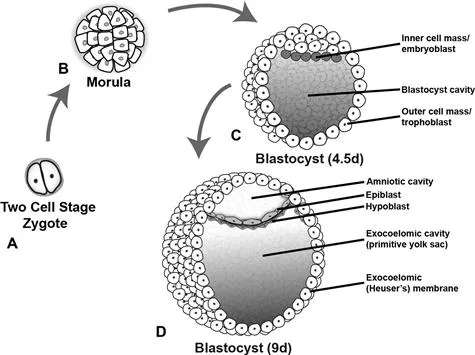
Fig. 1.1 Development of the bilaminar disc. The two-cell stage (A) is reached approximately 30 h after fertilization and the zygote eventually forms a 16-cell morula 2 days later (B). Inner and outer cell masses form at this time during a process referred to as compaction. The inner cell mass goes on to form the embryo, while the outer cell mass or trophoblast forms the placenta and extra-embryonic membranes. These masses become more apparent when the morula becomes a blastocyst 4.5 days after fertilization (C) and the blastocyst cavity develops. With further development the inner cell mass is known as the embryoblast. The blastocyst typically implants in the uterine mucosa 5–6 days after fertilization. The bilaminar disc forms within the blastocyst during the second week of development when the amniotic cavity develops within the epiblast (D). The constituent layers of the bilaminar disc are the epiblast (primitive ectoderm, lining the amniotic cavity) and the hypoblast (primitive endoderm, lining the primitive yolk sac).
In the second week of development, the inner cell mass, now known as the embryoblast, separates into two distinct cell layers, the hypoblast and the epiblast, which form the endoderm and ectoderm respectively. A second cystic cavity then develops adjacent to the epiblast. These layers thus form a bilaminar disc sandwiched between two cavities; the hypoblast lines the blastocyst cavity (primitive yolk sac) while the epiblast lines the developing amniotic cavity.
Gastrulation and Hensen’s node
In the second week gastrulation occurs, which establishes the third germ layer, mesoderm (Fig. 1.2). Gastrulation begins with formation of the primitive streak in the caudal region of the epiblast. The cranial end of the primitive streak forms a thickening known variously as the primitive knot, the primitive node, or Hensen’s node. The primitive pit forms immediately posterior to the node and cells from the epiblast migrate here, invaginate, and then form intraembryonic endoderm and mesoderm.
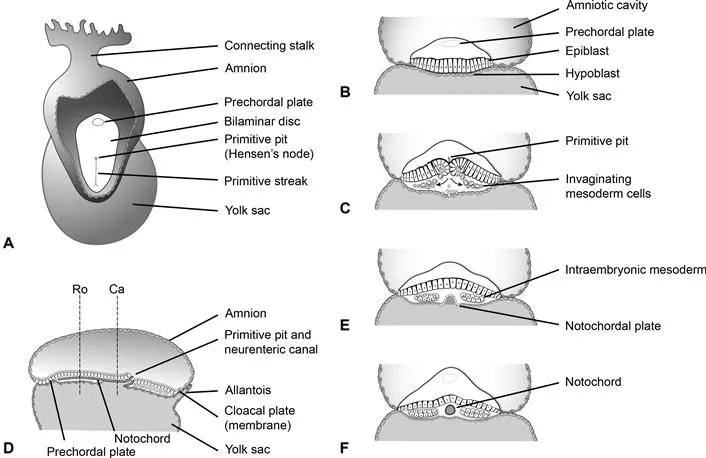
Fig. 1.2 Gastrulation and development of the notochord. At the end of the second week of development a thickening of cells forms in the caudal midline of the bilaminar germ disc, referred to as the primitive streak (A). The prechordal plate is visible at the rostral end of the disc and eventually develops into the buccopharyngeal membrane. (B) and (C) show coronal views through the bilaminar disc. Epiblast cells invaginate at the primitive pit and primitive streak creating the cells of the definitive endoderm as well as the mesoderm through the process of gastrulation. Prenotochordal cells invaginate during this process and migrate as far rostral as the prechordal plate. Initially they intercalate with the hypoblast forming the notochordal plate (E). The notochordal plate then detaches from the endoderm, and forms a tube referred to as the definitive notochord (F). (E) and (F) are coronal views looking rostral from planes Ro and Ca shown in (D), which is a mid-sagittal section through the embryo at 17d postfertilization. The neurenteric canal is a temporary communication between the amniotic cavity and yolk sac believed to play a central role in many malformations of the spine and spinal cord.
The primitive node migrates caudally as gastrulation progresses, and although it typically regresses and forms the caudal eminence or end bud after migration to the sacrococcygeal area, it is deserving of some further discussion. Hensen’s node secretes morphogens such as fibroblast growth factor (FGF), sonic hedgehog (Shh) and retinoic acid (RA), playing key roles in neural induction and patterning which will be discussed in detail. In this fashion, Hensen’s node establishes the longitudinal axis, polarity and right–left sidedness within the embryo. It also participates in rostrocaudal specification along with paraxial mesoderm. Failure of Hensen’s node to regress can lead to formation of a sacrococcygeal teratoma.
Formation of the notochord
Another critical event occurring in the second week is the formation of the notochord (see Fig. 1.2). The notochord is a cylindrical structure derived from mesodermal cells which specifies the midline of the embryo, in addition to forming a rigid axis around which the embryo can develop. It also secretes inductive signals critical to the formation of the nervous system from the overlying ectoderm.
Prenotochordal cells which form the notochord migrate in from the primitive streak, and move rostrally toward the prechordal plate (future buccopharyngeal membrane) to form the notochordal process, a precursor of the notochord. The notochordal process initially intercalates with the hypoblast to form the notochordal plate. At this time an important transitory communication between the amniotic cavity and yolk sac forms which is known as the neurenteric canal. This canal is of great significance to spine and spinal cord maldevelopment, as it is currently believed to play a critical role in numerous malformations such as neurenteric cysts and split cord malformations (Pang and Dias, 1992), as will be discussed.
Almost immediately after the notochordal plate forms and directly contacts the yolk sac, it separates from the endoderm, moves slightly dorsally and re-forms a cord of cells running along the rostrocaudal axis of the embryo’s midline. Despite its embryological significance, few notochord remnants persist in the adult. These cells make up the nucleus pulposus at the center of the intervertebral disc and notochord remnants are also believed to give rise to chordomas and notochordal rests (Kyriakos et al., 2003).
Primary neurulation
The central nervous system begins to develop in the third week postfertilization and the process begins with neurulation (Figs 1.3, 1.4). At the outset of neurulation, the notochord induces a subset of ectodermal cells to differentiate into neural precursor cells, forming a columnar epithelium referred to as the neural plate. Primary neurulation occurs when the neural plate folds and closes to form the neural tube.
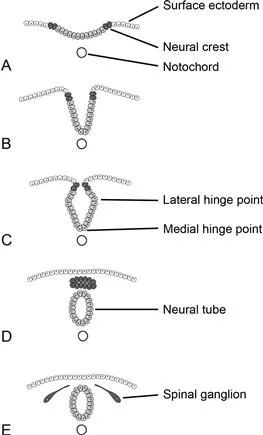
Fig. 1.3 Primary neurulation. The neural tube and cells of the neural crest are derived from surface ectoderm, forming a columnar epithelium referred to as the neural plate, as a result of induction by the notochord. Neural crest cells initially reside lateral to those that will form the neural tube (A). Folding internalizes these cells (B). Medial and lateral hinge points serve to anchor the neural tube, facilitating this folding (C). The medial hinge point is also known as the floor plate. The neural crest cells separate and form a mass dorsal to the neural tube (D). They later migrate to form dorsal root ganglia and many important cells types within the embryo (E).
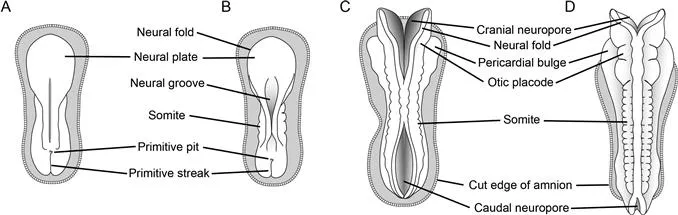
Fig. 1.4 Closure of the neural tube. Dorsal views of the embryo are shown with the amnion removed. (A) An 18-day-old embryo with a prominent neural plate is about to undergo primary neurulation. (B) At 20 days post‐fertilization, somites begin to appear and the neural folds begin to meet and fuse; this proc...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- Handbook of Clinical Neurology 3rd Series
- Foreword
- Preface
- List of contributors
- Chapter 1. Development and maturation of the spinal cord
- Chapter 2. Anatomy and biomechanics of the spinal column and cord
- Chapter 3. Clinical diagnosis and prognosis following spinal cord injury
- Chapter 4. Clinical neurophysiology in the prognosis and monitoring of traumatic spinal cord injury
- Chapter 5. Concepts of aging with paralysis
- Chapter 6. Advanced MRI strategies for assessing spinal cord injury
- Chapter 7. Advances in the management of spinal cord and spinal column injuries
- Chapter 8. Spinal cord injuries in children and adolescents
- Chapter 9. The changing landscape of spinal cord injury
- Chapter 10. Surgical treatments to restore function control in spinal cord injury
- Chapter 11. Advances in the rehabilitation management of acute spinal cord injury
- Chapter 12. Spasticity
- Chapter 13. Disordered cardiovascular control after spinal cord injury
- Chapter 14. New concepts in the prevention of pressure sores
- Chapter 15. Functional electrical stimulation for bladder, bowel, and sexual function
- Chapter 16. Evidence-based therapy for recovery of function after spinal cord injury
- Chapter 17. Functional electrical stimulation
- Chapter 18. Spinal cord stimulation
- Chapter 19. Promoting optimal functioning in spinal cord injury
- Chapter 20. The changing field of rehabilitation
- Chapter 21. The longitudinal spinal cord injury
- Chapter 22. Post-traumatic syringomyelia
- Chapter 23. Post-traumatic deformity
- Chapter 24. Spinal cord injury clinical trials translational process, review of past and proposed acute trials with reference to recommended trial guidelines
- Chapter 25. Translating preclinical approaches into human application
- Chapter 26. Translational spinal cord injury research
- Chapter 27. Brain–machine interfaces and transcranial stimulation
- Chapter 28. Approaches to repairing the damaged spinal cord: overview
- Chapter 29. Limiting spinal cord injury by pharmacological intervention
- Chapter 30. Harmful and beneficial effects of inflammation after spinal cord injury
- Chapter 31. Defeating inhibition of regeneration by scar and myelin components
- Chapter 32. Realizing the maximum potential of Schwann cells to promote recovery from spinal cord injury
- Chapter 33. Repair of central nervous system lesions by transplantation of olfactory ensheathing cells
- Chapter 34. Cell transplantation
- Chapter 35. Gene therapy, neurotrophic factors and spinal cord regeneration
- Chapter 36. Scaffolds to promote spinal cord regeneration
- Chapter 37. Molecular target discovery for neural repair in the functional genomics era
- Chapter 38. Combination therapies
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Spinal Cord Injury by Joost Verhaagen,John W. McDonald in PDF and/or ePUB format, as well as other popular books in Medicine & Neurology. We have over 1.5 million books available in our catalogue for you to explore.