
eBook - ePub
Pre-Examination Procedures in Laboratory Diagnostics
Preanalytical Aspects and their Impact on the Quality of Medical Laboratory Results
- 424 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Pre-Examination Procedures in Laboratory Diagnostics
Preanalytical Aspects and their Impact on the Quality of Medical Laboratory Results
About this book
The preanalytical phase is an important component of Laboratory medicine and errors arising in this phase affect the validity of laboratory results. In this book physicians and clinical staff have access to valuable information about the current preanalytical variables and factors (patient preparation, sample collection, handling and processing before analysis).
Tools to learn more effectively

Saving Books

Keyword Search

Annotating Text

Listen to it instead
Information
___
1. General Part
1.1 Introduction and History of the Preanalytical Phase
1.1.1 Introduction
It is extremely important to obtain adequate sample, ensure its appropriate transport and maintain proper storage before medical laboratories can perform diagnostic analysis [1]. Thirtyfive years after the introduction of the term preanalytical phase [2], it is now appropriate to summarize the existing knowledge, terms and recommendations about all biological and technical aspects between the patient and laboratory, which need to be controlled in order to obtain accurate laboratory result.
Most important of the whole diagnostic process, but often not considered as part of the quality criteria, is the extralaboratory part of the total turnaround time (TTT or total TAT) from the medical question to the sample [3]. This has been shown to be the source of a majority of “laboratory errors” [4] and is included in the ISO/ EN/DIN 15189 requirements for quality and competence (ISO 2007). It was only recently that quality indicators of the preanalytical phase were harmonized [5] (see Chapters 8.1–8.3).
Guidelines and diagnostic pathways helped to improve the selection of type of sample, time and site of sampling and also the definition of reference intervals for clinical interpretation of results (see Chapter 1.2). Various biological and technical aspects can either influence the laboratory result in vivo or interfere with the analytical procedure, thus changing the result and causing misinterpretations (see Chapter 1.3). These influences include age and gender differences, circadian rhythms as well as food, herbs and drinking habits (see Chapters 3.1–3.4). In addition, drugs can either change analyte concentrations by their biological actions or interfere with the analytical method used (see Chapter 4.4). Interferences can also becaused by secondary changes in sample color and turbidity by lipaemia, hemolysis or icterus (see Chapters 4.1–4.3). The site of sampling determines the result by biological variables (see Chapters 2.1–2.8). Biological influences can be the cause of differences between serum and plasma (see Chapter 2.6). Besides, anticoagulants, additives or contamination can also cause interferences.
The sampling is performed by varied professionals worldwide. The technical procedure including patient and sample identification, sampling techniques and storage and transport conditions are described in separate chapters (Chapters 5.1–5.3 and Chapter 6.1). An additional list provides information on the present state of knowledge about analyte stability in samples and their temperature dependence as well as stabilizing additives (see 10, Annex).
When a sample reaches the laboratory, several processes are mandatory before the requested analysis is performed (see Chapter 8.2). Individual aspects for the various analytes are discussed separately (on hemostaseology, hematology, acid–base state, biochemical and immunological analytes as well as therapeutic drug monitoring, microbiology and biobanking: see Chapters 7.1–7.7). Auditing and internal and external quality assurance covering all the facets of preanalytical phase are explained in Chapters 8.1–8.3, opening the discussion on unresolved questions [6].
Hopefully, this booklet contributes to the increasing awareness of the multiple aspects of laboratorial diagnostic function. Also, the editors wish to thank all the authors for their competent contributions; they have made this volume an updated component of the ongoing progress in medical treatment.
1.1.2 The long history prior to the appearance of the term preanalytical phase
Table 1.1.1: History of technical products and procedures involved in preanalytical phase
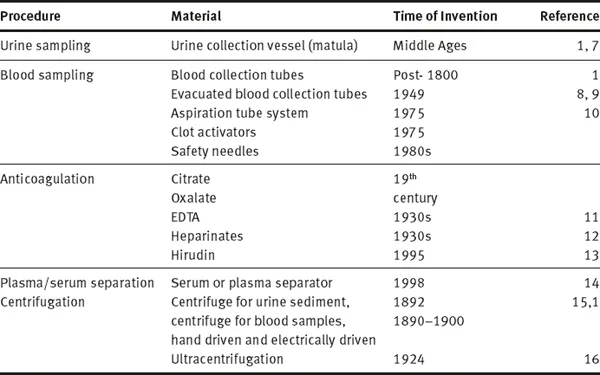
Definition and present state of concepts of preanalytical phase that emerged would not have been possible without more than hundred years of history of the preanalytical phase preceding it. Since chemical and microscopic analysis of body fluids improved medical diagnosis, the type of sample, time of sampling as well as the preparation of patients have been part of the method description. In addition, many inventions and technical improvements contributed to the standards that we use today. Table 1.1.1 summarizes some of the preanalytical techniques and inventions, which although existing since many decenniums are still considered standardized prerequisites for preanalytical procedures. Among the first preanalytical materials documented is the special urine sample container, the “matula” (Fig. 1.1.1), which based on Galen’s humoral theory allowed to separate hypostases (sediment) from sublimia (floating matter) and nubes (cloud).

Fig. 1.1.1: Uroscopy showing the preanalytical (man carrying the vessel protecting the matula filled with urine), analytical (uroscopist looking through the urine) and postanalytical phases (hand pointing to the brain of the uroscopist and rolling papers waiting for the prophetic (!) interpretation. (From a wood cut of Steffen Arndes from Lübeck 1510–20; from [17]).
As seen in Fig. 1.1.1 (circa 1500 AD), this matula was carried by the patient to the uroscopist in a matching cylindrical basket, which could be closed with a lid and carried with a handle. The basket protected urine from sunlight that would change the color or turbidity of urine. It was well known that without this protective basket urine was too unstable to get true informations about the color, consistency and contents.
Three hundred years later (post 1800 AD), only when newly developed chemical methods made the detection and quantitation of the constituents of body fluids possible, was blood used for chemical analysis. Initially, whole blood and/or serum, contained in blood tubes standing upright at room temperature for hours, were used as samples. The first use of a centrifuge was for separating urine sediment (Fig. 1.1.2). A hand-driven centrifuge rotor was used to spin two tubes with urine to examine sediment under microscope [15]. This remained a standardized procedure until electrically driven centrifuges with higher speed were available for blood centrifugation from 1920 onwards (although mentioned by Stenbeck in 1892 [15]). Ultimately, the invention of ultracentrifuge facilitated the separation of molecules according to molecular weight and specific weight [16]. Anticoagulants to preserve samples were used in the second half of the 19th century (citrate and oxalate, later EDTA and heparinates and recently hirudin), while standardized EDTA plasma was only possible after the 1940s [11]. These anticoagulants were added to a vessel before letting the blood flow into it and could only be roughly quantitated. Only with the invention of vacuum tubes and aspiration tubes [9, 10] standard volumes and anticoagulant concentrations were achieved. Years later, serum or plasma separators and additives stimulating coagulation in plastic tubes were introduced.

Fig. 1.1.2: Hand driven centrifuge used to form urine sediment.
It is well known among clinical scientists that preparation of the patient for sampling (diet, prolonged fasting prior to sampling, posture before and during sampling, physical activity, etc.), time and site of sampling (early morning versus afternoon, venous or capillary sample, etc.), choice of anticoagulant (serum or citrated-, EDTA- or heparinized blood, etc.), transport and storage (whole blood or serum, room temperature or cold, etc.) and centrifugation time and temperature exert their influence on laboratory results [18, 19]. However, the influences of these factors were not quantitated. Because their impact ...
Table of contents
- Cover
- Titel
- Imressum
- Preface
- Contents
- List of Contributing Authors
- 1 General Part
- 2 Types of Samples and Anatomic Site of Origin
- 3 Biological Variables Influencing Laboratory Results
- 4 Source and Nature of Interferences of Analytical Procedures
- 5 Sampling Materials and Techniques
- 6 Specimen Processing in the Preanalytical Phase
- 7 Preanalytical Variables and Rules in Specific Fields of Medical Laboratory Diagnostics
- 8 Quality Assurance and Auditing of the Preanalytical Phase
- 9 Annex: Samples and Stability of Analytes
- Fußnoten
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Pre-Examination Procedures in Laboratory Diagnostics by Walter G. Guder in PDF and/or ePUB format, as well as other popular books in Medicine & Biochemistry in Medicine. We have over one million books available in our catalogue for you to explore.