![]()
1.1Introduction
Type 2 diabetes (T2D) has evolved into a leading public health concern both for its high prevalence and devastating complications. Current global estimates of 422 million individuals affected by T2D represent a fourfold increase since 1980, with a continued rapid rise.1 The International Diabetes Federation (IDF) similarly quotes 451 million individuals affected aged 18–99 years in 2017 (8.4%), with a projected prevalence of 693 million or 9.9% by 2045.2 Interestingly, the rise in incidence comes at a time when accumulated evidence clearly demonstrates that T2D is a preventable disease.3 Prior to the 1900s, T2D and obesity were exceedingly rare, affecting two individuals per 100 000 population and 1 in 30 persons, respectively.4 The modern-day lifestyle has largely contributed to the overwhelming disease burden, in part due to the increasing availability of processed foods and a sedentary lifestyle facilitated by technology. Obesity, itself, confers a fourfold increase in risk of T2D, and represents the single greatest modifiable risk factor for development of the disease.5 In contrast to T2D, Type 1 diabetes (T1D) results from the loss of pancreatic β-cell production of insulin most often through autoimmune mechanisms.4 Though patients with T1D also benefit from dietary modification,5 strategies for T1D are beyond the scope of this chapter and book, which focus on insulin resistance syndromes. Given the extensive disease morbidity, mortality, and global healthcare expenditure, an intensive lifestyle intervention that includes a nutrient-rich dietary pattern, among other modifications, is vital for prevention and management of these chronic metabolic conditions.6,7
1.2Mechanisms of Insulin Resistance and Metabolic Syndrome
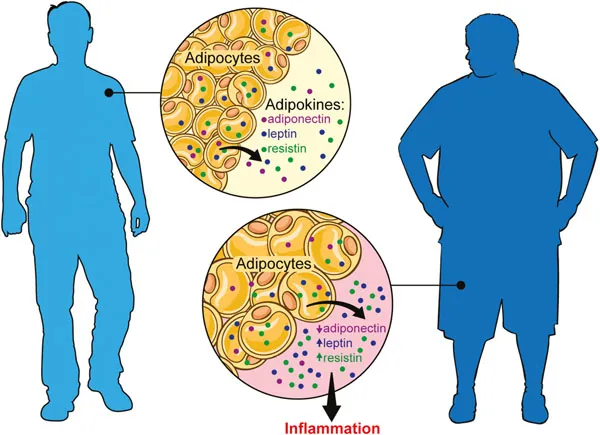
The presence of insulin resistance may be demonstrated in patients long before the development of T2D and is believed to serve as the main underlying driver of the disease process.8 Obesity predisposes to insulin resistance through a number of complex mechanisms, in part related to increased adipocyte size and quantity and to adipocyte dysfunction.9 Adipose tissue, traditionally viewed as simple fat storage, is now more aptly recognized as an active endocrine organ that secretes adipokines, including adiponectin, leptin and resistin, which regulate multiple metabolic pathways involved in glucose homeostasis.10–12 The excess caloric intake associated with obesity overwhelms adipocyte mechanisms, resulting in dysregulated adipokine secretion, and ultimately a pro-inflammatory state.13 Enlarged adipocytes further encourage macrophage recruitment to the tissue, prompting chronic low-grade inflammation.14 Once established, insulin resistance behaves as a highly disruptive state to regulation of metabolism, exerting widespread effects that include raised glucagon activity during meals that results in inappropriate prandial hepatic glucose release, impaired glucagon-like peptide 1 (GLP-1) release, leptin resistance, reduced ghrelin and adiponectin activity, systemic endoplasmic reticulum stress, systemic inflammation, and pancreatic β-cell dysfunction which culminate clinically as T2D.15 Similar pathophysiologic mechanisms are observed in other conditions of insulin resistance, including the metabolic syndrome (MetS), hypertriglyceridemia, non-alcoholic hepatosteatosis, and polycystic ovarian syndrome (PCOS).
MetS represents a composite of clinical risk factors that predict insulin resistance, and cumulatively contribute to high risk for atherosclerotic cardiovascular disease (ASCVD).15 Though published classifications vary slightly, the National Cholesterol Education Program Adult Treatment Panel III defines MetS by the presence of three of the following five parameters: (1) increased abdominal girth (waist circumference >102 cm in men and >88 cm in women); (2) hypertension (systolic blood pressure >130 mmHg and diastolic blood pressure >85 mmHg); (3) elevated circulating triglycerides (>150 mg dL–1); (4) reduced circulating high-density lipoprotein (HDL) levels (<40 mg dL–1 in men and <50 mg dL–1 in women); and (5) elevations in serum glucose concentrations (fasting serum glucose >100 mg dL–1).16 Using these criteria, MetS was observed in 33% of adults in the US population in 2012, and demonstrates a rising prevalence worldwide.17 Due to overlapping pathophysiology, hepatosteatosis, seen in approximately 30% of US adults, and PCOS, observed in 7–10% of reproductive-age women, are also highly prevalent in modern populations.18,19
Dietary strategies leading to significant and sustained weight loss can successfully hamper the underlying pathophysiologic mechanisms and mitigate both the development and progression of syndromes of insulin resistance, including T2D and MetS, as well as hepatosteatosis and PCOS by extension (Figure 1.1).
1.3Prevention Trials
Several landmark trials support the hypothesis that lifestyle interventions leading to weight loss can substantially alter metabolic factors that predispose to T2D and MetS. The first, in 1997, the Da Qing Trial20 in China, involved 577 individuals with impaired glucose tolerance randomly assigned to either conventional lifestyle or one of three treatment groups consisting of dietary intervention, physical activity (PA), or both. All three intervention groups were associated with a significant reduction in the incidence of T2D as compared to control, with an incidence of 67.7%, 43.8%, 41.1%, and 46% with conventional lifestyle, dietary intervention, PA intervention, and both diet and PA interventions, respectively, providing early evidence for lifestyle intervention as a low-cost and effective method of preventing these challenging diseases.
The Finnish Diabetes Prevention Study21 (DPS) was another early, randomized trial to demonstrate a causative association between lifestyle modification and T2D prevention in high-risk individuals. The Finnish DPS consisted of 522 overweight adults aged 40–64 with impaired glucose tolerance randomized to either an intensive lifestyle group or a control group receiving standard of care dietary recommendations. With target weight loss of at least 5% body weight, the study showed a 58% risk reduction of developing T2D at 3 years, 43% at 7 years, and 32% reduction at 13-year follow-up compared to the control group. Marked improvement was also noted with respect to glycemic control and cholesterol levels with lifestyle intervention.
In a larger randomized trial of 3234 participants in the United States, the Diabetes Prevention Program22 (DPP) confirmed the findings of the Finnish DPS by further demonstrating the impact of an intensive lifestyle intervention for the prevention of T2D in high-risk populations. Results from the DPP showed a 58% reduction in the incidence of T2D with an intensive ...