eBook - ePub
The Molecular Nutrition of Amino Acids and Proteins
A Volume in the Molecular Nutrition Series
- 368 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
The Molecular Nutrition of Amino Acids and Proteins
A Volume in the Molecular Nutrition Series
About this book
The Molecular Nutrition of Amino Acids and Proteins provides an in-depth look at the involvement and role of amino acids and proteins in molecular nutrition. Editor Dominique Dardevet has assembled a collection of chapters written by leading researchers and top professors that provide the reader with a comprehensive understanding of amino acids and proteins.
The book provides an introduction to the fundamentals of amino acids and proteins as well as the composition of food. It then delves into the molecular biology of the cell and genetic machinery and its function. The Molecular Nutrition of Amino Acids and Proteins also features reference guides for terms and bullet-point summaries, making it readily accessible to novices while still providing the most up-to-date and detailed information that experienced researchers need.
- Provides a gentle introduction to the subject by first addressing nutritional information and then building in molecular aspects, clearly establishing fundamental information for the reader
- Facilitates reader comprehension by including succinct summary points in each chapter
- Contains a glossary of definitions that allows readers to easily reference terms
- Provides both a deep and broad understanding of the subject by containing overviews as well as detail-focused chapters
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Section II
Cellular Aspects of Protein and Amino Acids Metabolism in Anabolic and Catabolic Situations
Outline
Chapter 6
Amino Acids and Exercise
Molecular and Cellular Aspects
C. McGlory and S.M. Phillips, Exercise Metabolism Research Group, Department of Kinesiology, McMaster University, Hamilton, ON, Canada
Abstract
Resistance exercise and amino acid feeding are potent stimuli for the accrual of skeletal muscle, and as such, are often used as a means to increase muscle mass in athletes or combat muscle loss in disease states. In response to resistance exercise and amino acid feeding there is a profound stimulation of muscle protein synthesis (MPS) that is underpinned by a series of protein kinase phosphorylation events. Our understanding as to how different types and compositions of amino acids impact the muscle protein synthetic response to resistance exercise while evolving is incomplete at present. Moreover, the study of how amino acids alter the activation of key signaling molecules known to stimulate MPS remains in its infancy. The aim of this chapter is to concisely summarize our current understanding of how exercise and protein nutrition alter skeletal muscle protein turnover and both the cellular and molecular level. We also provide some insight as to potential areas for future research.
Keywords
Muscle protein synthesis; resistance exercise; leucine; mTORC1
6.1 Introduction
Skeletal muscle is a critical and often unappreciated organ that not only supports human locomotion but its mass is also a robust predictor for all-cause mortality (Kallman et al., 1990; Metter et al., 2002). Furthermore, given that the human body is 45% skeletal muscle by mass, sustaining skeletal muscle mass throughout life is essential for promoting both athletic performance and longevity. Despite its importance, several of the physiological mechanisms that regulate the size of human muscle mass have only recently been elucidated, and many are still unknown. What is known is that the interaction between amino acid feeding-induced changes in rates of muscle protein synthesis (MPS) and rates of muscle protein breakdown (MPB) ultimately dictate net muscle protein balance (NPB) (Phillips et al., 1997). However, the composition, dose, timing, and daily distribution of protein intake (and subsequent effect on aminoacidemia) throughout the day that induces optimal MPS remains a topic of intense research and debate. Moreover, how these variables influence the cellular and molecular regulators that mediate amino acid-induced increases in MPS, particularly those involved in translation initiation and elongation, remain largely unknown. The aim of this chapter is to provide a critical evaluation of our current understanding of how amino acid ingestion, mainly in the form of intact protein sources, following loading (resistance exercise) influences MPS at both the cellular and molecular level. For the purposes of concision and relevance to the human model, data reported primarily derived from human studies will be cited but, where appropriate, work from other experimental models will be introduced to substantiate points of discussion.
6.2 Regulation of the Size of Human Muscle Mass
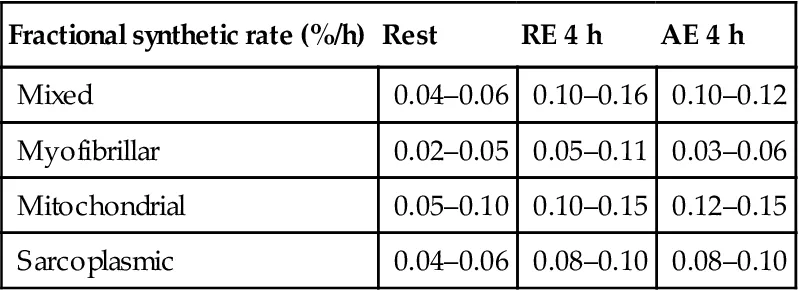
The size of human skeletal muscle mass is dependent upon the coordinated interaction between changes in rates of MPS and MPB (Phillips et al., 1997). The basal and fasted state rates of MPB are known to exceed those of MPS and thus, skeletal muscle net protein balance (NPB=MPS minus MPB) is negative. Over two decades ago, using stable isotopic infusions, it was demonstrated that a mixed macronutrient meal was capable of increasing rates of MPS above rates of MPB and that the amino acids contained within the meal were primarily responsible for this increase (Bennet et al., 1990; Rennie et al., 1982). However, after approximately 2–3 h rates of MPS are known to decline to postabsorptive levels until the consumption of the next amino acid-containing meal (Areta et al., 2013; Atherton et al., 2010). Interestingly, performing exercise, particularly resistance exercise, prior to consuming amino acids has been shown to potentiate rates of MPS (Phillips et al., 2012; Witard et al., 2009). It is this potentiation by resistance exercise of feeding-induced increases in MPS that is responsible for the hypertrophic phenotype observed with resistance exercise training and protein feeding over time (Fig. 6.1). Importantly, not all amino acids exert the same stimulatory effect of MPS in skeletal muscle either at rest or following exercise, and identifying the relevant amino acids, optimal amino acid/protein dose and/or composition of amino acids to promote gains in muscle size and function is of great scientific interest.
6.3 Exercise Mode
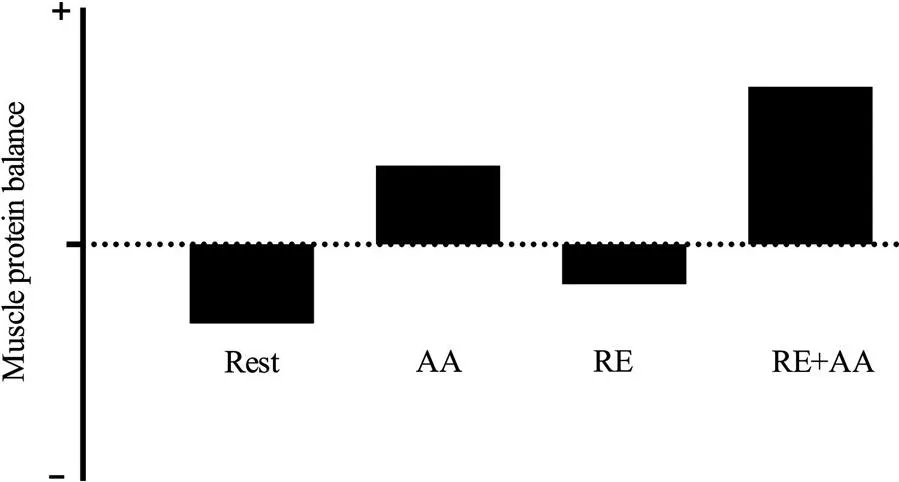
While resistance exercise will remain the focus of this chapter it is important to recognize that even aerobic exercise can stimulate MPS (Carraro et al., 1990; Harber et al., 2010) and even result in protein accretion (Harber et al., 2009, 2012). However, the fraction of muscle proteins that are predominantly turned over in response to exercise is specific to the intensity and duration of the exercise bout, as well as the training status of the individual (Burd et al., 2010; Wilkinson et al., 2008). For example, in an untrained state, resistance exercise stimulates an increase in both myofibrillar and mitochondrial MPS; however, in the trained state, resistance exercise only stimulates an increase in myofibrillar MPS (Table 6.1). Endurance exercise on the other hand stimulates an increase in mitochondrial MPS in both the trained and untrained state, but does not result in a stimulation of myofibrillar MPS (Wilkinson et al., 2008). These differences are important considerations when evaluating the efficacy of a given exercise or nutritional stimulus to alter protein turnover as the assessment of mixed MPS can mask any potential protein fraction-specific differences in rates of synthesis (Kim et al., 2005). It is also known that high intensity intermittent exercise training (HIIT), a stimulus that could be considered a “hybrid” of both resistance and endurance exercise, has the capacity to stimulate both myofibrillar and mitochondrial MPS (Bell et al., 2015; Scalzo et al., 2014). Although ingestion of protein (and the subsequent aminoacidemia) is thought to influence the adaptive response to all modes of exercise, the effect is most profound following resistance exercise (Cermak et al., 2012, 2013). As a result, the continuing theme of this chapter will be how protein/amino acid feeding alters the protein synthetic response to resistance exercise with some discussion relating to the role of endurance and HIIT exercise.
Table 6.1
Examples from the literature of rates of muscle protein synthesis captured 4 h following either resistance exercise or endurance exercise
| Fractional synthetic rate (%/h) | Rest | RE 4 h | AE 4 h |
| Mixed | 0.04–0.06 | 0.10–0.16 | 0.10–0.12 |
| Myofibrillar | 0.02–0.05 | 0.05–0.11 | 0.03–0.06 |
| Mitochondrial | 0.05–0.10 | 0.10–0.15 | 0.12–0.15 |
| Sarcoplasmic | 0.04–0.06 | 0.08–0.10 | 0.08–0.10 |
Resistance exercise (RE) values obtained from Burd et al. (2010), and aerobic exercise (AE) values obtained from Wilkinson et al. (2008). All mitochondrial protein turnover rates were obtained from Wilkinson et al. (2008).
6.4 Protein Type
A major independent variable that drives MPS is the digestion rate and subsequent aminoacidemia of ingested proteins. This factor can largely be influenced by the quality of intact proteins as defined by various scoring systems such as the Digestible Indispensible Amino Acid Score (for an expanded review see van Vliet et al., 2015). The most widely studied categories of dietary isolated protein are whey, casein and soy, all of which have differing digestion and absorption kinetics as well as amino acid composition. In this regard, isolated whey and soy proteins, since both are acid-soluble, are relatively rapidly digested resulting in a heightened but transient hyperaminoacidemia (Devries and Phillips, 2015). Soy protein however, contains a greater proportion of nonessential amino acids than whey protein (Mahe et al., 1996). Casein in its micellar form (as it exists in milk) is acid insoluble and coagulates in the stomach, which slows transit time into the intestine and thus results in an aminoacidemia that is smaller in amplitude but greater in duration than whey (Boirie et al., 1997).
To date, the influence of protein type on exercise-induced increases in MPS has been understudied. One study has shown that postprandial rates of MPS are greater following the consumption of whey and soy as compared with casein (Tang et al., 2009). Additionally, whey protein consumption has been shown to be superior to both soy and casein for the purposes of stimulating MPS following resistance exercise (Burd et al., 2012; Tang et al., 2009). The superiority of whey to stimulate rates of MPS following resistance exercise is attributed to the high essential amino acid, specifically leucine (Churchward-Venne et al., 2014a) content, the influence of which will be discussed later in this chapter. Another hypothesis is that the rapid increase in blood amino acid concentrations associated with whey protein drives MPS. Indeed, in one study where whey protein was consumed either as a 25 g bolus immediately following resistance exercise or as 10 individual 2.5 g “pulse” drinks separated by 20 min, it ...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- List of Contributors
- Preface
- Section I: General and Introductory Aspects
- Section II: Cellular Aspects of Protein and Amino Acids Metabolism in Anabolic and Catabolic Situations
- Section III: Cellular and Molecular Actions of Amino Acids in non Protein Metabolism
- Section IV: Dietary Amino Acid and Protein on Gene Expression
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access The Molecular Nutrition of Amino Acids and Proteins by Dominique Dardevet in PDF and/or ePUB format, as well as other popular books in Tecnología e ingeniería & Nutrición, dietética y bariatría. We have over 1.5 million books available in our catalogue for you to explore.