
eBook - ePub
MRI in Practice
Catherine Westbrook, John Talbot
This is a test
Buch teilen
- English
- ePUB (handyfreundlich)
- Über iOS und Android verfügbar
eBook - ePub
MRI in Practice
Catherine Westbrook, John Talbot
Angaben zum Buch
Buchvorschau
Inhaltsverzeichnis
Quellenangaben
Über dieses Buch
MRI in Practice continues to be the number one reference book and study guide for the registry review examination for MRI offered by the American Registry for Radiologic Technologists (ARRT). This latest edition offers in-depth chapters covering all core areas, including: basic principles, image weighting and contrast, spin and gradient echo pulse sequences, spatial encoding, k-space, protocol optimization, artefacts, instrumentation, and MRI safety.
- The leading MRI reference book and study guide.
- Now with a greater focus on the physics behind MRI.
- Offers, for the first time, equations and their explanations and scan tips.
- Brand new chapters on MRI equipment, vascular imaging and safety.
- Presented in full color, with additional illustrations and high-quality MRI images to aid understanding.
- Includes refined, updated and expanded content throughout, along with more learning tips and practical applications.
- Features a new glossary.
MRI in Practice is an important text for radiographers, technologists, radiology residents, radiologists, and other students and professionals working within imaging, including medical physicists and nurses.
Häufig gestellte Fragen
Wie kann ich mein Abo kündigen?
Gehe einfach zum Kontobereich in den Einstellungen und klicke auf „Abo kündigen“ – ganz einfach. Nachdem du gekündigt hast, bleibt deine Mitgliedschaft für den verbleibenden Abozeitraum, den du bereits bezahlt hast, aktiv. Mehr Informationen hier.
(Wie) Kann ich Bücher herunterladen?
Derzeit stehen all unsere auf Mobilgeräte reagierenden ePub-Bücher zum Download über die App zur Verfügung. Die meisten unserer PDFs stehen ebenfalls zum Download bereit; wir arbeiten daran, auch die übrigen PDFs zum Download anzubieten, bei denen dies aktuell noch nicht möglich ist. Weitere Informationen hier.
Welcher Unterschied besteht bei den Preisen zwischen den Aboplänen?
Mit beiden Aboplänen erhältst du vollen Zugang zur Bibliothek und allen Funktionen von Perlego. Die einzigen Unterschiede bestehen im Preis und dem Abozeitraum: Mit dem Jahresabo sparst du auf 12 Monate gerechnet im Vergleich zum Monatsabo rund 30 %.
Was ist Perlego?
Wir sind ein Online-Abodienst für Lehrbücher, bei dem du für weniger als den Preis eines einzelnen Buches pro Monat Zugang zu einer ganzen Online-Bibliothek erhältst. Mit über 1 Million Büchern zu über 1.000 verschiedenen Themen haben wir bestimmt alles, was du brauchst! Weitere Informationen hier.
Unterstützt Perlego Text-zu-Sprache?
Achte auf das Symbol zum Vorlesen in deinem nächsten Buch, um zu sehen, ob du es dir auch anhören kannst. Bei diesem Tool wird dir Text laut vorgelesen, wobei der Text beim Vorlesen auch grafisch hervorgehoben wird. Du kannst das Vorlesen jederzeit anhalten, beschleunigen und verlangsamen. Weitere Informationen hier.
Ist MRI in Practice als Online-PDF/ePub verfügbar?
Ja, du hast Zugang zu MRI in Practice von Catherine Westbrook, John Talbot im PDF- und/oder ePub-Format sowie zu anderen beliebten Büchern aus Medicina & Tecnología y suministros médicos. Aus unserem Katalog stehen dir über 1 Million Bücher zur Verfügung.
Information
1
Basic principles
- Introduction
- Atomic structure
- Motion in the atom
- MR-active nuclei
- The hydrogen nucleus
- Alignment
- Net magnetic vector (NMV)
- Precession and precessional (Larmor) frequency
- Precessional phase
- Resonance
- MR signal
- The free induction decay (FID) signal
- Pulse timing parameters
After reading this chapter, you will be able to:
- Describe the structure of the atom.
- Explain the mechanisms of alignment and precession.
- Understand the concept of resonance and signal generation.
INTRODUCTION
The basic principles of magnetic resonance imaging (MRI) form the foundation for further understanding of this complex subject. It is important to grasp these ideas before moving on to more complicated topics in this book.
There are essentially two ways of explaining the fundamentals of MRI: classically and via quantum mechanics. Classical theory (accredited to Sir Isaac Newton and often called Newtonian theory) provides a mechanical view of how the universe (and therefore how MRI) works. Using classical theory, MRI is explained using the concepts of mass, spin, and angular momentum on a large or bulk scale. Quantum theory (accredited to several individuals including Max Planck, Albert Einstein, and Paul Dirac) operates at a much smaller, subatomic scale and refers to the energy levels of protons, neutrons, and electrons. Although classical theory is often used to describe physical principles on a large scale and quantum theory on a subatomic level, there is evidence that all physical principles are explained using quantum concepts [1]. However, for our purposes, this chapter mainly relies on classical perspectives because they are generally easier to understand. Quantum theory is only used to provide more detail when required.
In this chapter, we explore the properties of atoms and their interactions with magnetic fields as well as the mechanisms of excitation and relaxation.
ATOMIC STRUCTURE
All things are made of atoms. Atoms are organized into molecules, which are two or more atoms arranged together. The most abundant atom in the human body is hydrogen, but there are other elements such as oxygen, carbon, and nitrogen. Hydrogen is most commonly found in molecules of water (where two hydrogen atoms are arranged with one oxygen atom; H2O) and fat (where hydrogen atoms are arranged with carbon and oxygen atoms; the number of each depends on the type of fat).
The atom consists of a central nucleus and orbiting electrons (Figure 1.1). The nucleus is very small, one millionth of a billionth of the total volume of an atom, but it contains all the atom’s mass. This mass comes mainly from particles called nucleons, which are subdivided into protons and neutrons. Atoms are characterized in two ways.
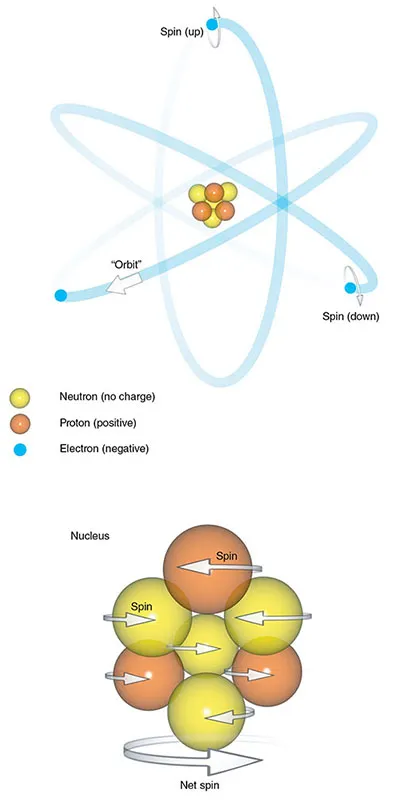
Figure 1.1 The atom.
- The atomic number is the sum of the protons in the nucleus. This number gives an atom its chemical identity.
- The mass number or atomic weight is the sum of the protons and neutrons in the nucleus.
The number of neutrons and protons in a nucleus is usually balanced so that the mass number is an even number. In some atoms, however, there are slightly more or fewer neutrons than protons. Atoms of elements with the same number of protons but a different number of neutrons are called isotopes.
Electrons are particles that spin around the nucleus. Traditionally, this is thought of as analogous to planets orbiting around the...