This established textbook offers a one-stop, comprehensive coverage of air pollution, all in an easy-reading and accessible style. The fourth edition, broadly updated and developed throughout, includes a brand-new chapter providing a broader overview to the topic for general reading, and presents fresh materials on air pollution modelling, mitigation and control, tailored to the needs of both amateur and specialist users. Retaining a quantitative perspective, the covered topics include: gaseous and particulate air pollutants, measurement techniques, meteorology and modelling, area sources, mobile sources, indoor air, effects on plants, materials, humans and animals, impact on climate change and ozone profiles and air quality legislations. This edition also includes a final chapter covering a suite of sampling and laboratory practical experiments that can be used for either classroom teachings, or as part of research projects. As with previous editions, the book is aimed to serve as a useful reading resource for upper-level undergraduate and postgraduate courses specialising in air pollution, with dedicated case studies at the end of each chapter, as well as a list of revision questions provided at the end as a complementary section.

eBook - ePub
Air Pollution
Measurement, Modelling and Mitigation, Fourth Edition
- 696 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
BiowissenschaftenSubtopic
UmweltwissenschaftChapter 1
Overview of air pollution
1.1 WHAT IS AIR POLLUTION?
Our Earth is uniquely gifted with an envelope of chemicals that enables life-supporting activities – commonly known as air. At least 3000 different chemicals have been identified in air samples. However, it can safely be assumed that the sum total of these chemicals is at least equal to those that have been originally produced on the Earth, plus additional ones formed by their subsequent reactions.
Air pollution is defined as ‘the presence of substances in the atmosphere that can cause adverse effects to man and the environment’. It is a term used to describe any unwanted chemicals or other materials that contaminate the air we breathe, resulting in the reduction of its life-supporting qualities (commonly referred to as air quality). In principle, an air pollutant is any chemical species that exceeds the concentrations or characteristics of the natural constituents of air (discussed in Section 1.5.2). However, strictly speaking, a pollutant is defined as a substance that is potentially harmful to the health or well-being of human, animal or plant life, or to the ecological systems.
Pollution (in the general sense) was defined in the Tenth Report of the Royal Commission on Environmental Pollution as:
The introduction by man into the environment of substances or energy liable to cause hazard to human health, harm to living resources and ecological systems, damage to structure or amenity or interference with legitimate use of the environment.
This is a very broad definition that includes many types of pollution that we shall not cover in this book, yet it contains some important ideas. Note that by this definition, chemicals such as sulphur dioxide from volcanoes or methane from the decay of natural vegetation are not counted as pollution, but sulphur dioxide from coal-burning or methane from rice-growing are pollution. Radon, a radioactive gas that is a significant natural hazard in some granitic areas, is not regarded as pollution as it does not arise from people’s activities. The boundaries become fuzzier when we are dealing with natural emissions that are influenced by our actions – for example, there are completely natural biogenic emissions of terpenes from forests, and our activities in changing the local patterns of land use have an indirect effect on these emissions. Air pollution, as discussed in this book, is the solid, liquid or gaseous material emitted into the air from stationary or mobile sources, moving subsequently through an aerial path, and perhaps being involved in chemical or physical transformations before eventually being returned to the surface. The material has to interact with something before it can have any environmental impacts. This interaction may be, for example, with other molecules in the atmosphere (photochemical formation of ozone from hydrocarbons), with electromagnetic radiation (by greenhouse gas molecules), with liquid water (the formation of acid rain from sulphur dioxide), with vegetation (the direct effect of ozone), with mineral surfaces (soiling of buildings by particles) or with animals (respiratory damage by acidified aerosol). Pollution arising from human activities is called anthropogenic, while those having other natural origins (animals or plants) is said to be biogenic. Air pollution has been identified as a major problem in modern society, although in its conventional form of smoke and fumes, its origins date back to the Middle Ages, being closely associated with the Industrial Revolution and the use of coal. Originally, air pollution included only substances from which environmental damage was anticipated because of their toxicity or their specific capacity to damage organisms or structures. However, in the last decade, the scope has been widened to include substances such as chlorofluorocarbons, ammonia or carbon dioxide that have more general environmental impacts. Table 1.1 provides the most recently updated list of local and transboundary air pollutants along with the details of the international and legislative directives that regulate them in the UK. Most of these standards and the legislation are described in greater detail in Chapter 14.
In recent years there has been huge focus on nitrogen oxides (NOx) – a combination of two gases, nitrogen dioxide (NO2) and nitric oxide (NO). In fact, it is the NO2 that affects human health (hence, subjected to more stringent statutory emission limit guidelines set by the European Union, World Health Organization [WHO], United States Environmental Protection Agency [USEPA], etc., as discussed in Chapter 14). The directly emitted NO2 (also known as primary NO2) immediately influences the ambient concentration. However, our atmosphere is a dynamic lab and the NO eventually reacts with ozone (O3) to form additional NO2 (known as secondary NO2), thereby increasing their effective human health concerns.
Table 1.1 Local and transboundary air pollutants along with their regulatory international and UK legislative directives
Air pollutants | Directive |
PM – PM10, PM2.5, NOx, O3, SO2, PAHs, Benzene, 1,3-butadiene, CO, Pb | Air Quality Strategy |
SO2, NH3, NOx, NMVOC | National Emissions Ceilings Directive (NECD) |
SO2, NH3, NOx, NMVOC, Heavy Metals, POPs | Convention on Long-Range Transboundary Air Pollutants (CLRTAP) |
91 compounds including: CH4, CO, CO2 HFCs, N2O, SF6, NH3, NMVOC, NOx, PFCs, SOx, CFCs, As, Cd, Cr, Cu, Hg, Ni, Pb, Zn, PM10, Benzene, HCl, HF, PAHs, PCBs, PCDD, PCDF, Gamma HCH, PCP, HCB | European Pollutant Release and Transfer Register (E-PRTR), which succeeds the European Pollutant Emission Register (EPER) |
SO2, NOx, CO, VOCs, metals, dust, asbestos, chlorine and its compounds | Integrated Pollution Prevention and Control (IPPC) |
SOx, NOx, PM | Large Combustion Plants Directive (LCPD) |
Dust (PM), HCl, HF, SO2, NOx, Heavy metals, Dioxins and Furans, CO | Waste Incineration Directive (WID) |
VOCs | Solvent Emissions Directive |
VOC | Paints Directive |
SO2 | The Sulphur Contents of Liquid Fuels Directive |
VOC | Petrol vapour recovery |
SO2, NOx, PM, Lead, Benzene, CO, Ozone, PAH, Cadmium, Arsenic, Nickel, Mercury | EU Air Quality Directives |
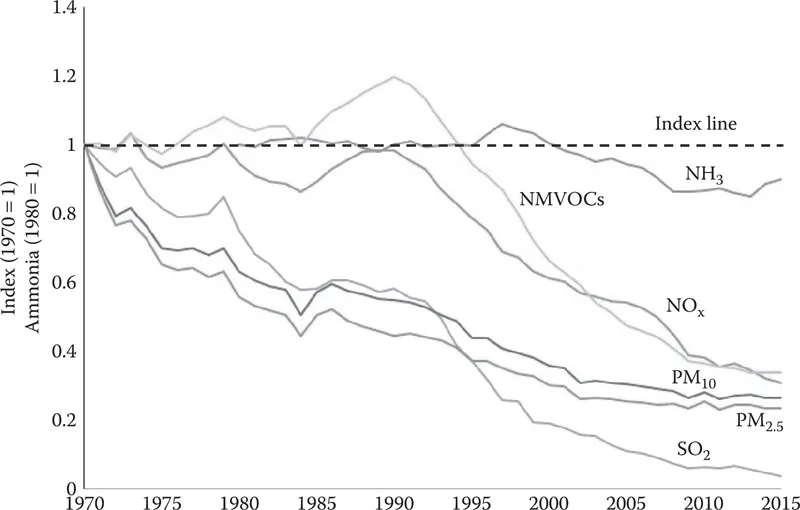
Figure 1.1 Historical trends in UK emissions of key air pollutants between 1970 and 2015. (From NAEI, UK emissions of air pollutants, National Atmospheric Emissions Inventory, http://naei.beis.gov.uk/overview/ap-overview, 2016.)
In the UK, there has been long-term decrease in emissions of all of the air pollutants compared to the historical releases during the second half of the twentieth century. Figure 1.1 presents historical trends in the overall emissions of a number of air pollutants in the UK between 1970 and 2015 in terms of a reference index, which is acquired by normalising the reported annual totals by the UK government with the reference emissions ...
Table of contents
- Cover
- Half Title
- Title Page
- Copyright Page
- Table of Contents
- Preface
- Authors
- 1 Overview of air pollution
- 2 Gaseous air pollutants
- 3 Particulate matter
- 4 Area sources
- 5 Mobile sources
- 6 Ambient air quality
- 7 Indoor air quality
- 8 Air pollution modelling
- 9 Air pollution control and mitigation
- 10 Air pollution impacts
- 11 Air pollution and climate change
- 12 Air pollution impacts on ozone
- 13 Noise and light pollution
- 14 Air quality standards and legislations
- 15 Air quality experiments
- Review questions
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Air Pollution by Abhishek Tiwary,Ian Williams in PDF and/or ePUB format, as well as other popular books in Biowissenschaften & Umweltwissenschaft. We have over 1.5 million books available in our catalogue for you to explore.