
eBook - ePub
Chiroptical Spectroscopy
Fundamentals and Applications
Prasad L. Polavarapu
This is a test
Partager le livre
- 430 pages
- English
- ePUB (adapté aux mobiles)
- Disponible sur iOS et Android
eBook - ePub
Chiroptical Spectroscopy
Fundamentals and Applications
Prasad L. Polavarapu
Détails du livre
Aperçu du livre
Table des matières
Citations
À propos de ce livre
This book details chiroptical spectroscopic methods: electronic circular dichroism (ECD), optical rotatory dispersion (ORD), vibrational circular dichroism (VCD), and vibrational Raman optical activity (VROA). For each technique, the text presents experimental methods for measurements and theoretical methods for analyzing the experimental data. It also includes a set of experiments that can be adopted for undergraduate teaching laboratories. Each chapter is written in an easy-to-follow format for novice readers, with necessary theoretical formalism in appendices for advanced readers.
Foire aux questions
Comment puis-je résilier mon abonnement ?
Il vous suffit de vous rendre dans la section compte dans paramètres et de cliquer sur « Résilier l’abonnement ». C’est aussi simple que cela ! Une fois que vous aurez résilié votre abonnement, il restera actif pour le reste de la période pour laquelle vous avez payé. Découvrez-en plus ici.
Puis-je / comment puis-je télécharger des livres ?
Pour le moment, tous nos livres en format ePub adaptés aux mobiles peuvent être téléchargés via l’application. La plupart de nos PDF sont également disponibles en téléchargement et les autres seront téléchargeables très prochainement. Découvrez-en plus ici.
Quelle est la différence entre les formules tarifaires ?
Les deux abonnements vous donnent un accès complet à la bibliothèque et à toutes les fonctionnalités de Perlego. Les seules différences sont les tarifs ainsi que la période d’abonnement : avec l’abonnement annuel, vous économiserez environ 30 % par rapport à 12 mois d’abonnement mensuel.
Qu’est-ce que Perlego ?
Nous sommes un service d’abonnement à des ouvrages universitaires en ligne, où vous pouvez accéder à toute une bibliothèque pour un prix inférieur à celui d’un seul livre par mois. Avec plus d’un million de livres sur plus de 1 000 sujets, nous avons ce qu’il vous faut ! Découvrez-en plus ici.
Prenez-vous en charge la synthèse vocale ?
Recherchez le symbole Écouter sur votre prochain livre pour voir si vous pouvez l’écouter. L’outil Écouter lit le texte à haute voix pour vous, en surlignant le passage qui est en cours de lecture. Vous pouvez le mettre sur pause, l’accélérer ou le ralentir. Découvrez-en plus ici.
Est-ce que Chiroptical Spectroscopy est un PDF/ePUB en ligne ?
Oui, vous pouvez accéder à Chiroptical Spectroscopy par Prasad L. Polavarapu en format PDF et/ou ePUB ainsi qu’à d’autres livres populaires dans Ciencias físicas et Química industrial y técnica. Nous disposons de plus d’un million d’ouvrages à découvrir dans notre catalogue.
Informations
1
Polarized Light
Interaction between electromagnetic radiation and matter is the basis of a broad research area that is generally categorized as spectroscopy. When the matter under consideration is made up of chiral molecules (see Chapter 2), a special branch of spectroscopy, labeled as chiroptical spectroscopy, becomes pertinent. The words “chiroptical spectroscopy” can be construed to mean the study of chiral systems using optical spectroscopy methods. To understand the principles of chiroptical spectroscopy, it is necessary to gain insight into the properties of electromagnetic radiation, chiral molecules, and the interaction between them. These concepts form the basis for the starting chapters in this book.
1.1 Introduction to Light
Visible light represents electromagnetic radiation that can be detected with the human eye. This statement automatically raises thoughts about “invisible” light. To distinguish between visible and invisible light, we need to consider the wave properties of electromagnetic radiation. A wave (see Figure 1.1a) is associated with periodic oscillatory variation of its height (referred to as amplitude) in time. Two identical points in a wave constitute one cycle. For example, two consecutive crest points, two consecutive trough points, or three consecutive zero points constitute one cycle or one wave. If an observer stands at one point and watches the waves pass by in time, the number of cycles passed in a given second is labeled as frequency (ν). The unit of frequency is cycles/second (or simply s−1), which is designated as hertz (Hz; 1 Hz = s−1). A cosine wave with unit amplitude is represented by the function cos2πν t, where t stands for time in seconds. This cosine wave is shown in Figure 1.1a, where the definition of a cycle is depicted. The alternate representations of frequency of oscillation are wavelength and wavenumber. If a stationary wave is held along the z axis (see Figure 1.1b) and the distance between three consecutive zero points (or between two crests or between two troughs) of that wave is measured, then that distance is labeled as wavelength and represented by symbol λ [expressed as meters/cycle or simply as meters (m)]. This definition of wavelength is shown in Figure 1.1b, where a wave with unit amplitude, represented by the cosine function cos2πz/λ, is depicted. The product of wavelength and frequency of a wave is equal to the speed of that wave c, which is expressed as m·s−1:
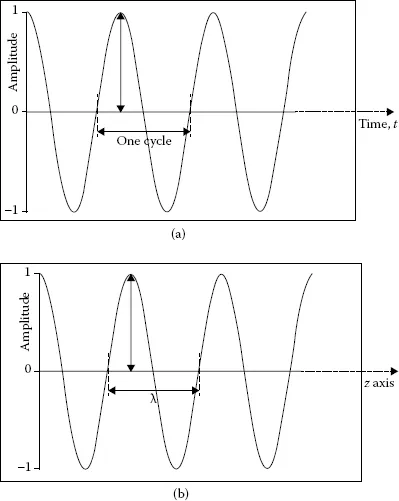
FIGURE 1.1
(a) Depiction of the amplitude and cycle of an oscillating cosine wave, cos 2πνt, with unit amplitude as a function of time. (b) Depiction of the amplitude and wavelength λ of an oscillating cosine wave, cos (2πz/λ), propagating along the z axis with unit amplitude.
(1.1) |
If an electromagnetic wave passes through a medium with refractive index nm, then the speed of electromagnetic wave changes as cm = c/nm. Then, Equation 1.1 modifies as cm = λmν, where λm is the wavelength of the wave in that medium (Levine 2009). For the current purposes, consider the waves propagating in vacuum, where nm = 1. Just as the number of cycles per second is labeled as frequency, the number of waves per unit length is labeled as wavenumber (). Since one wave occupies a length of λ, wavenumber is equal to the reciprocal of wavelength. Therefore, = 1/λ, and its units are expressed in m−1. Thus, an alternate form of Equation 1.1 is as follows:
(1.2) |
The wavelength λ (or equivalently frequency ν or wavenumber ) of a wave determines the properties of that wave. Human eyes respond to the electromagnetic waves of wavelength between ~400 and 800 nm (1 nm = 10−9 m), and this portion of electromagnetic radiation is referred to as the visible light. The different colors that human eyes can recognize (violet, indigo, blue, green, yellow, orange, and red, abbreviated as VIBGYOR) are the components of the visible light. The portions of electromagnetic radiation with wavelengths greater or less than the wavelengths in the visible light range are invisible to the human eye.
From here onward, the word “light” will be used to represent electromagnetic radiation (regardless of whether it is visible or invisible to the human eyes). The speed of light in vacuum is a constant, 2.99792458 × 108 m·s−1. Wavelength and frequency of a light wave are related through Equations 1.1 and 1.2. The different wavelength ranges of electromagnetic radiation constitute different portions of the electromagnetic “spectrum.” Selected portions of electromagnetic spectrum and their names are summarized in Table 1.1.
Wavelength is expressed in nanometers (nm) for the ultraviolet–visible region and micrometers (μm) for the infrared region. Wavenumber is usually expressed in cm−1. For example, a light wave with 200-nm wavelength represents ultraviolet light and corresponds to a wavenumber of 50,000 cm−1 and a frequency of 1.5 × 1015 Hz .
TABLE 1.1
Selected Regions of E...